Alpha Decay - Definition, Examples, Quiz, FAQ, Trivia
Discover how some atoms release energy by emitting alpha particles
What is Alpha Decay?
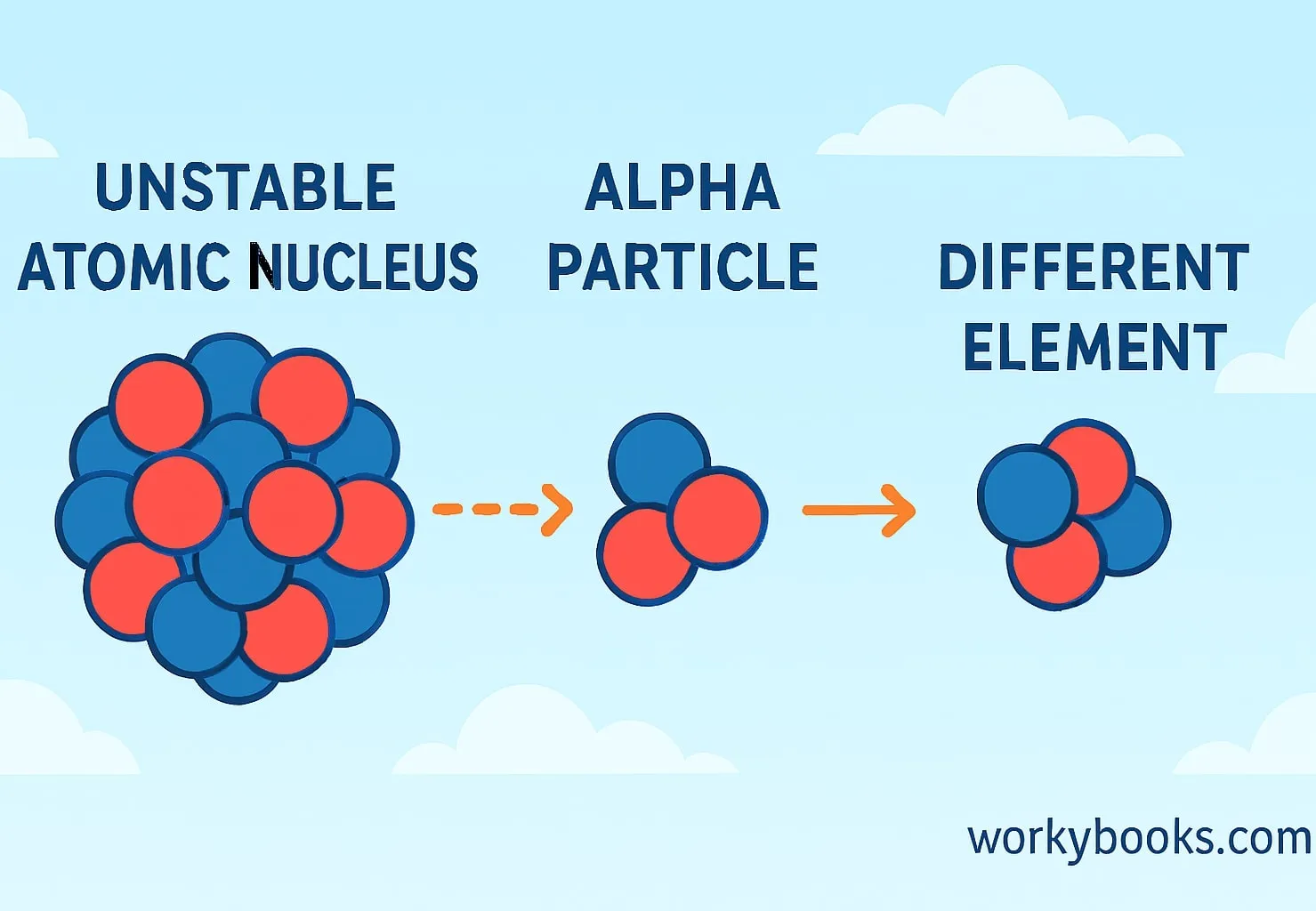
Alpha decay is a type of radioactive decay where an unstable atomic nucleus releases an alpha particle to become more stable. An alpha particle is made of two protons and two neutrons, which is the same as a helium nucleus.
Think of alpha decay like a crowded bus where some people decide to get off to make more space. The bus (the atomic nucleus) becomes less crowded and more stable after some passengers (the alpha particle) leave.
This process happens naturally in some heavy elements like uranium and radium. These elements have too many particles in their nucleus, making them unstable. By emitting an alpha particle, they transform into a different, more stable element.
Science Fact!
Alpha particles were first identified by Ernest Rutherford in 1899 through experiments with radioactive materials.
How Alpha Decay Works
Alpha decay happens when the strong nuclear force that holds protons and neutrons together isn't strong enough to overcome the repulsive forces between positively charged protons. Here's how this process works:
Unstable Nucleus
A heavy nucleus has too many protons and neutrons
Alpha Formation
Two protons and two neutrons group together
Emission
The alpha particle is ejected from the nucleus
New Element
The original atom becomes a different element
Energy Release
Energy is released during the process
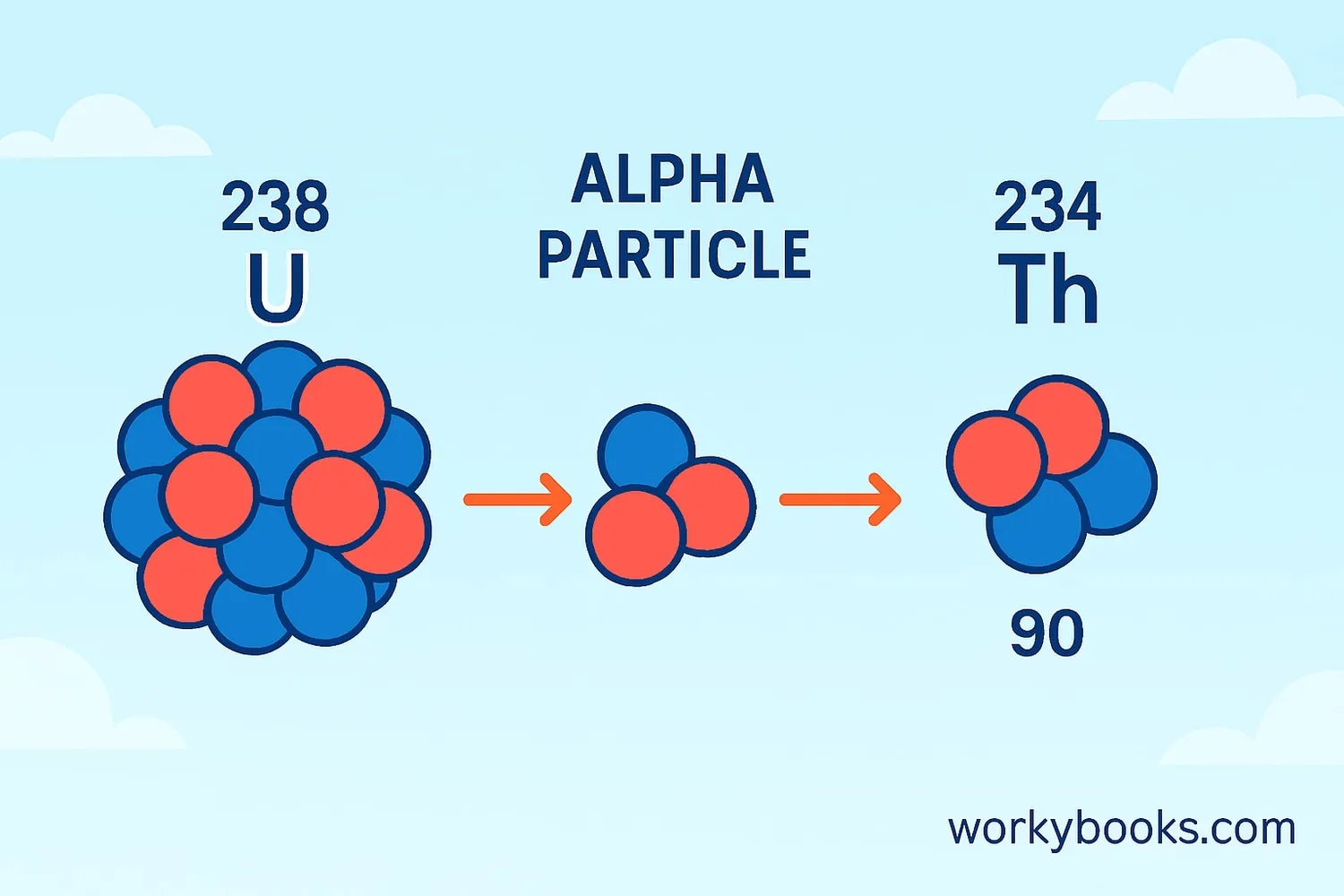
The mathematical representation of alpha decay shows how the atomic number and mass number change:
Nuclear Transformation!
During alpha decay, the atomic number decreases by 2 and the mass number decreases by 4, creating a completely different element!
Why Alpha Decay is Important
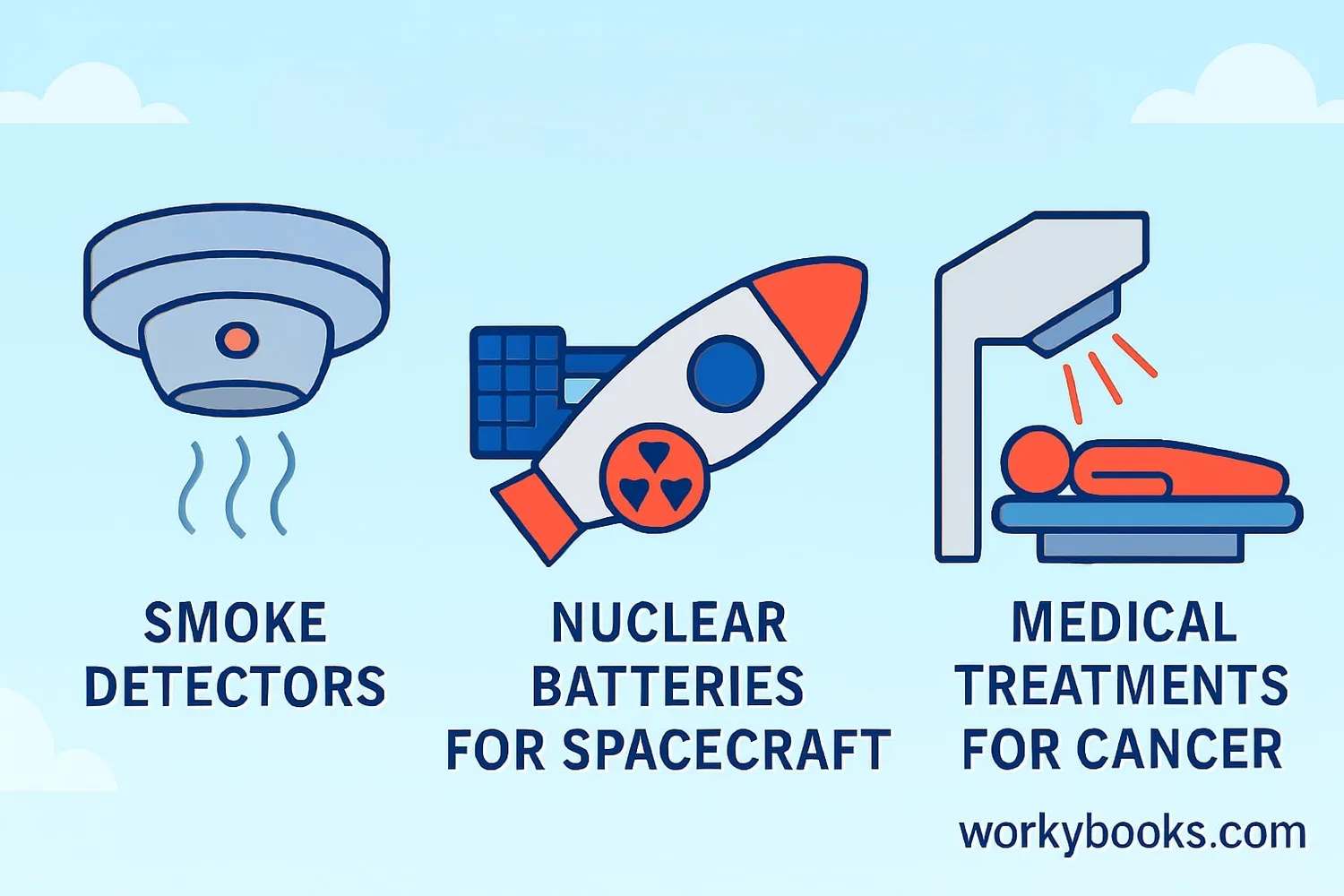
Alpha decay plays important roles in both nature and technology. Here's why it matters:
Smoke Detectors
Alpha particles ionize air in smoke detectors, allowing them to detect smoke quickly
Space Power
Radioisotope thermoelectric generators use alpha decay to power spacecraft
Medical Uses
Alpha emitters are used in targeted cancer treatments
Without understanding alpha decay, we wouldn't have:
• Effective smoke detectors that save lives
• Long-lasting power sources for space missions
• Certain cancer treatments that target diseased cells
• Methods to date ancient rocks and artifacts
Alpha decay also helps scientists understand how elements formed in stars and how our planet has changed over billions of years!
Alpha Decay Quiz
Test your alpha decay knowledge with this quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to some common questions about alpha decay:
Alpha Decay Trivia
Discover some amazing facts about alpha decay!
Natural Radiation
Alpha particles from radon gas in soil and rocks account for about half of the natural background radiation that humans are exposed to annually.
Space Exploration
The Voyager spacecraft, launched in 1977, are still operating using power from plutonium-238 radioisotope thermoelectric generators that rely on alpha decay.
Historical Use
Before the dangers were fully understood, alpha-emitting radium was used in glow-in-the-dark paints for watch dials in the early 20th century.
Scientific Discovery
Alpha decay experiments were crucial to Rutherford's discovery of the atomic nucleus in 1911, which revolutionized our understanding of atomic structure.

