Concentration - Definition, Examples, Quiz, FAQ, Trivia
Discover how scientists measure amounts of substances in solutions
What is Concentration?
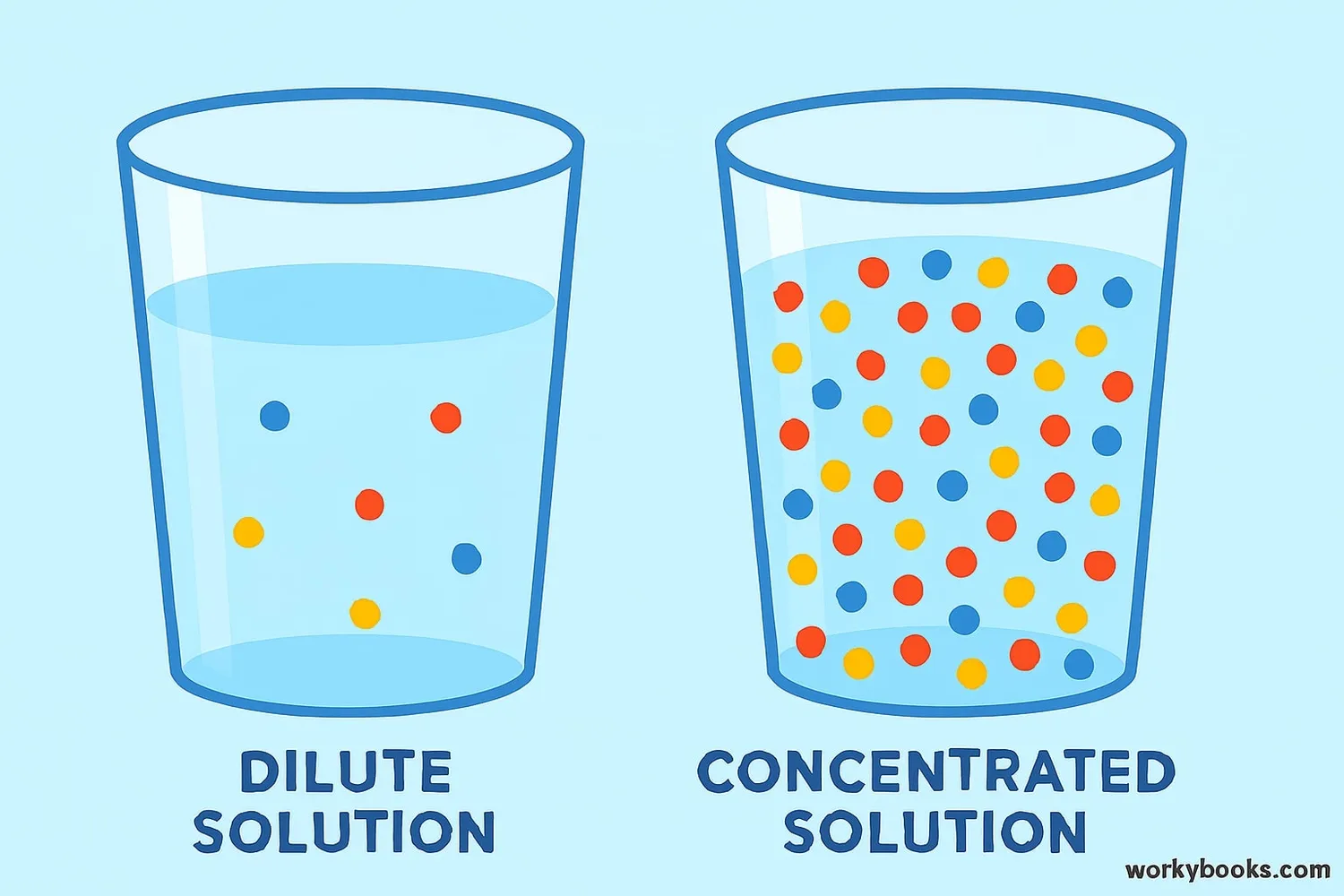
Concentration tells us how much of one substance is mixed with another substance. Imagine making lemonade:
• If you put just a little lemon juice in a lot of water, you have a dilute solution
• If you put a lot of lemon juice in a little water, you have a concentrated solution
Concentration is important because it helps scientists know exactly how much of a substance is in a mixture. This is essential for cooking, medicine, environmental science, and many other fields!
Science Fact!
Our taste buds can detect concentration differences! That's why we can taste when lemonade is too weak or too strong.
Measuring Concentration

Scientists use different units to measure concentration depending on what they're studying. Here are the most common ways:
| Unit | Symbol | Definition | Example |
|---|---|---|---|
| Molarity | M | Moles of solute per liter of solution | 1 M salt water = 58.5g salt in 1 liter water |
| Molality | m | Moles of solute per kilogram of solvent | 1 m salt water = 58.5g salt in 1 kg water |
| Parts per million | ppm | 1 part solute per 1 million parts solution | 1 ppm = 1 drop in 50 liters |
| Parts per billion | ppb | 1 part solute per 1 billion parts solution | 1 ppb = 1 drop in 20 swimming pools |
Molarity (M)
Most common unit for solutions in chemistry labs
Molality (m)
Used when temperature changes affect volume
Parts per million
Used for very dilute solutions like pollutants
Parts per billion
Used for extremely tiny amounts like vitamins in food
Real World Connection!
Water quality tests measure pollutants in ppm. Safe drinking water has less than 10 ppm of certain chemicals.
Why Concentration Matters

Concentration is important in many areas of science and daily life:
Medicine
Correct concentrations ensure medicines are safe and effective
Cooking
Recipes require specific concentrations of ingredients
Environment
Scientists measure pollution concentrations in water and air
Understanding concentration helps us:
• Create safe medicines and vaccines
• Keep our drinking water clean
• Make delicious food with consistent flavors
• Develop new materials and technologies
• Monitor environmental health
Concentration measurements are essential for scientific experiments, manufacturing, and quality control in countless industries!
Concentration Quiz
Test your knowledge about concentration with this quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to some common questions about concentration:
Science Trivia
Discover some amazing facts about concentration!
Super Tasters
Some people are "supertasters" who can detect concentrations 100 times weaker than average people. They have more taste buds and are extra sensitive to flavors!
Ocean Saltiness
The average salt concentration in ocean water is about 35,000 ppm. That's like 35 grams of salt in every liter of water - enough to fill a salt shaker!
Pollution Detection
Scientists can detect pollutants at concentrations as low as 1 ppb - that's equivalent to finding one specific person in all of China!
Space Chemistry
In space, fluids behave differently, making concentration measurements challenging. NASA scientists have developed special tools to measure concentrations in microgravity.


