Isotopes - Definition, Examples, Quiz, FAQ, Trivia
Discover how atoms of the same element can be different!
What are Isotopes?
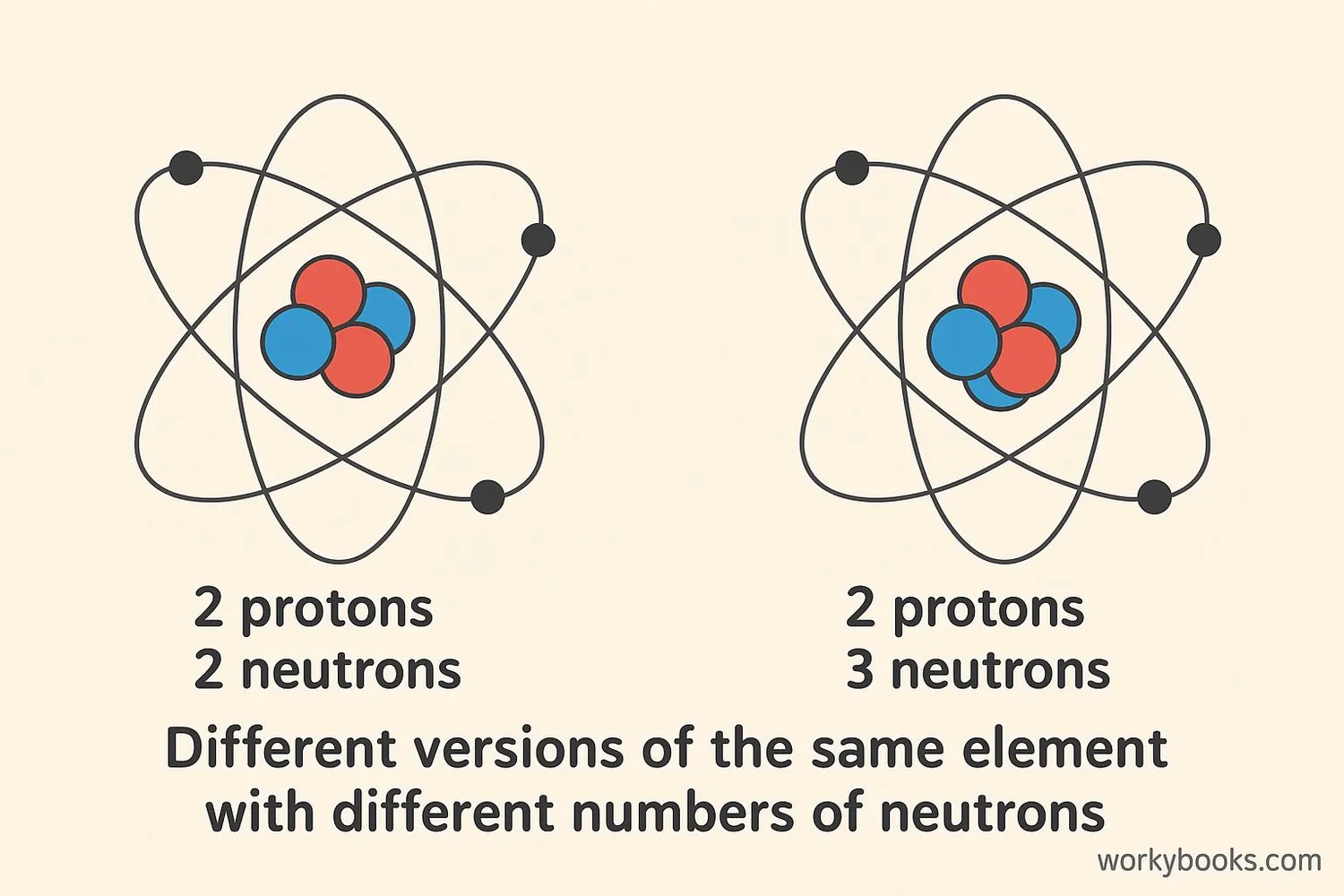
Isotopes are different versions of the same element. They have the same number of protons but different numbers of neutrons in their atoms.
Think of elements like different types of building blocks. Just like you can have different versions of the same block (some heavier, some lighter), elements can have different versions too! These different versions are called isotopes.
Every atom has:
• Protons (positively charged particles)
• Neutrons (neutral particles with no charge)
• Electrons (negatively charged particles that orbit the nucleus)
Key Concept!
The atomic number (number of protons) defines the element, while the mass number (protons + neutrons) helps identify different isotopes.
Protium
Hydrogen-1
Mass Number: 1
Deuterium
Hydrogen-2
Mass Number: 2
Tritium
Hydrogen-3
Mass Number: 3
All three of these are hydrogen isotopes! They each have 1 proton (which makes them hydrogen), but different numbers of neutrons. Protium has no neutrons, deuterium has 1, and tritium has 2.
Types of Isotopes

There are two main types of isotopes: stable isotopes and radioactive isotopes (also called radioisotopes).
Stable Isotopes
These isotopes don't change over time. Their atoms stay the same and don't give off radiation.
Radioactive Isotopes
These isotopes are unstable. Over time, they break down and release energy in a process called radioactive decay.
Half-Life
This is the time it takes for half of the radioactive atoms in a sample to decay. Different radioisotopes have different half-lives.
Example: Carbon Isotopes
Carbon-12 (stable) - Makes up most of the carbon in our bodies
Carbon-14 (radioactive) - Used in dating ancient objects (half-life: 5,730 years)
Other important isotopes:
• Uranium-235: Used in nuclear power plants
• Cobalt-60: Used in cancer treatment
• Technetium-99m: Used in medical imaging
• Oxygen-18: Used in climate research
Uses of Isotopes
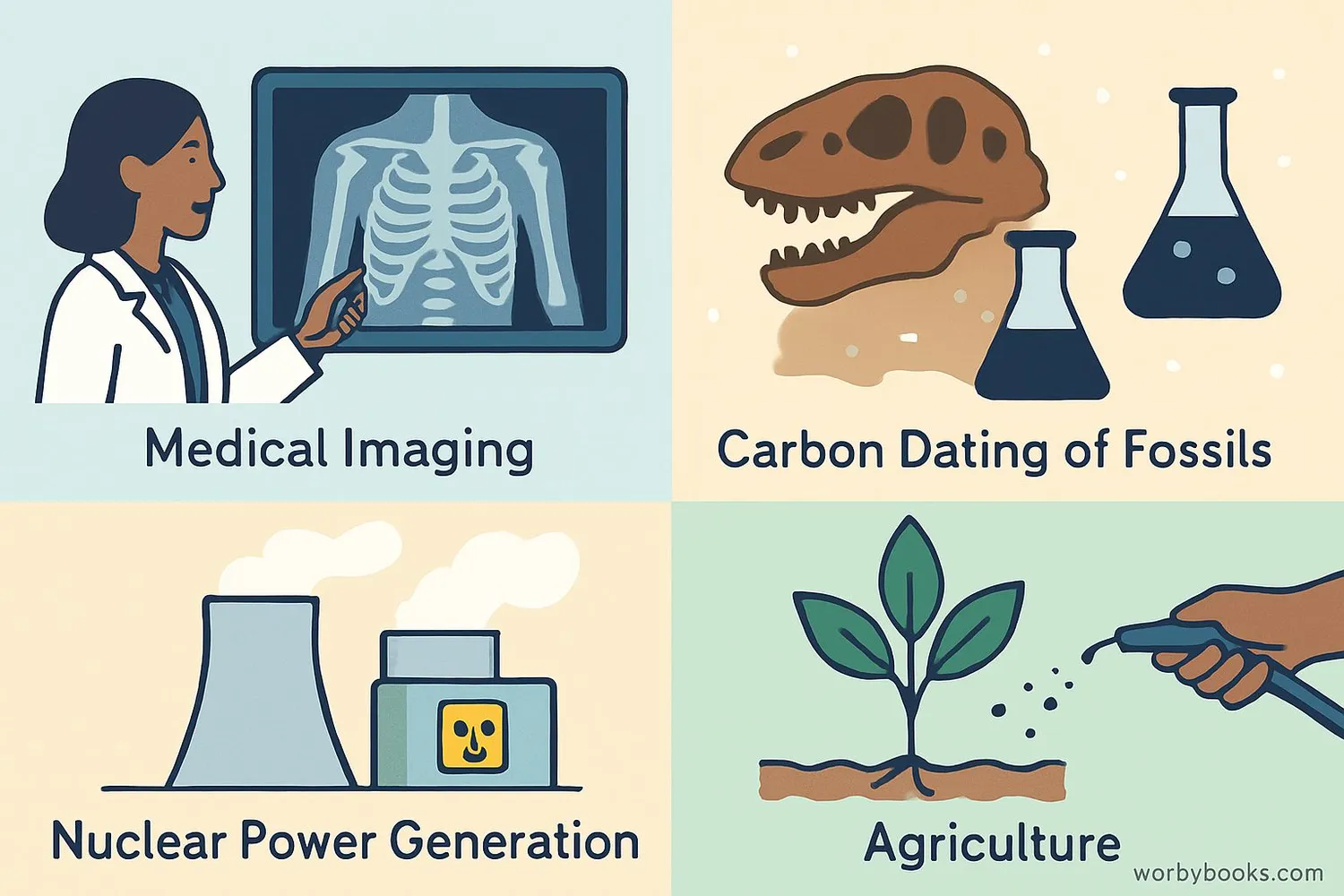
Isotopes have many important uses in science, medicine, industry, and our daily lives!
Medical Uses
Radioisotopes are used in medical imaging (like PET scans) and cancer treatment.
Carbon Dating
Scientists use Carbon-14 to determine the age of ancient artifacts and fossils.
Nuclear Power
Uranium-235 is used as fuel in nuclear power plants to generate electricity.
Agriculture
Radioisotopes help scientists develop better crops and understand plant growth.
Kinetic Isotope Effect
Scientists use isotopes to study chemical reactions. When an atom is replaced by its heavier isotope, the reaction might slow down. This "kinetic isotope effect" helps us understand how reactions work.
Isotopes Quiz
Test your isotope knowledge with this fun quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to some common questions about isotopes:
Fun Isotope Trivia
Discover some amazing facts about isotopes!
Heavy Water
Water made with deuterium (hydrogen-2) is called "heavy water" because it's about 10% heavier than regular water! It's used in some nuclear reactors.
Radioactive Bananas?
Bananas contain potassium-40, a naturally occurring radioactive isotope! But don't worry - you'd need to eat millions of bananas at once to get a harmful dose.
Space Power
Some spacecraft use radioisotope thermoelectric generators (RTGs) that convert heat from decaying plutonium-238 into electricity to power instruments in deep space.
Medical Marvel
Technetium-99m is used in about 85% of all nuclear medicine procedures worldwide - that's over 40 million procedures each year!


