Valence Electrons - Definition, Examples, Quiz, FAQ, Trivia
Discover how atoms bond and form molecules!
What Are Valence Electrons?
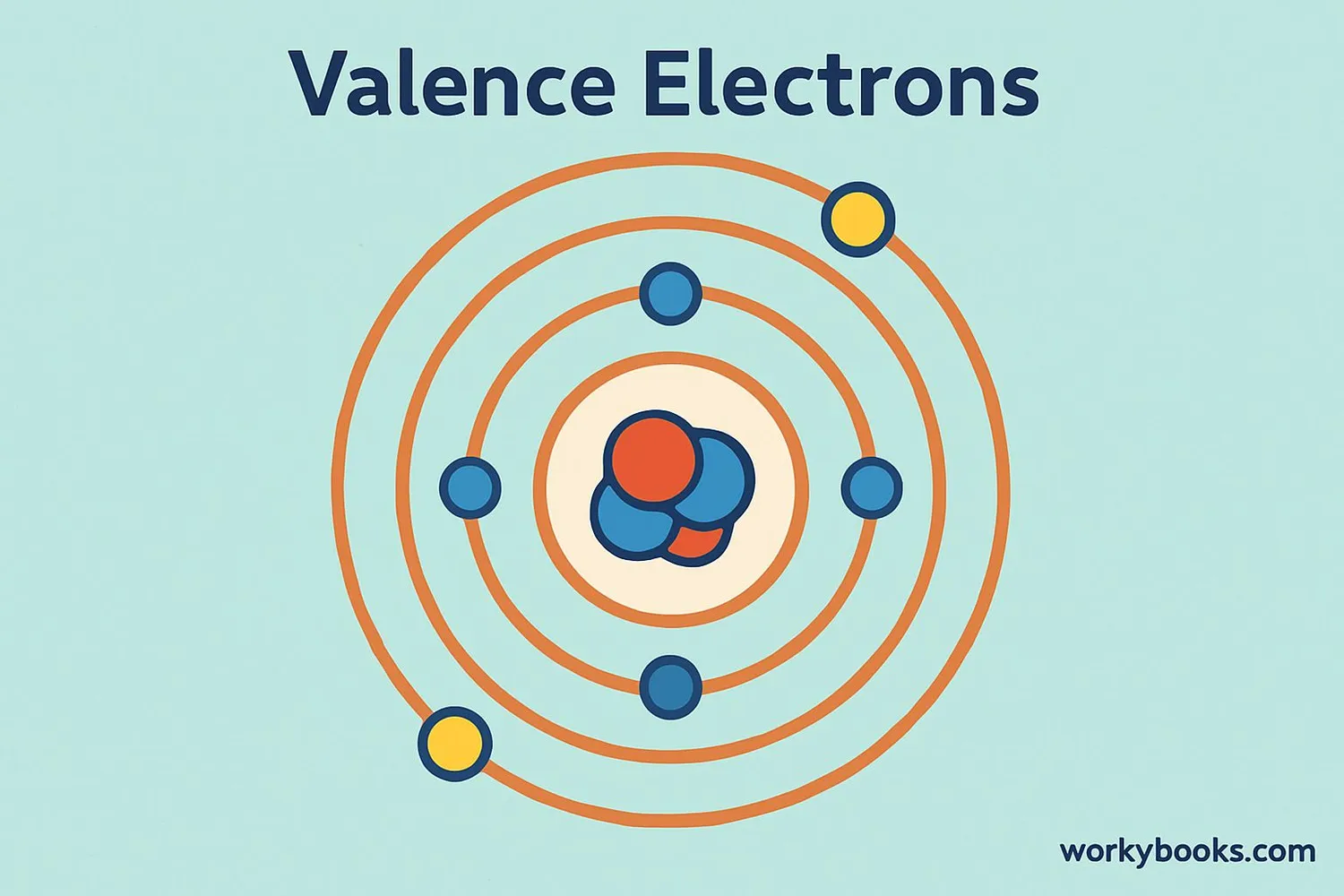
Valence electrons are the electrons in the outermost shell of an atom. These special electrons determine how atoms interact with each other to form chemical bonds and create molecules.
Think of atoms as tiny solar systems! The nucleus is the sun at the center, and electrons orbit around it in different shells. The electrons in the shell farthest from the nucleus are the valence electrons. These are the electrons that participate in chemical reactions.
Science Fact!
Atoms are most stable when they have 8 valence electrons (except hydrogen and helium which need only 2). This is called the octet rule.
Finding Valence Electrons
1
1
2
3
4
5
6
7
8
You can find how many valence electrons an atom has by looking at its position on the periodic table:
Group 1
1 valence electron
Group 2
2 valence electrons
Groups 13-18
3-8 valence electrons
For example:
• Sodium (Na) is in Group 1 → 1 valence electron
• Oxygen (O) is in Group 16 → 6 valence electrons
• Neon (Ne) is in Group 18 → 8 valence electrons
Electron Configuration
Electron configuration describes how electrons are arranged in an atom. The valence electrons are always in the highest energy level.
Chemical Bonding
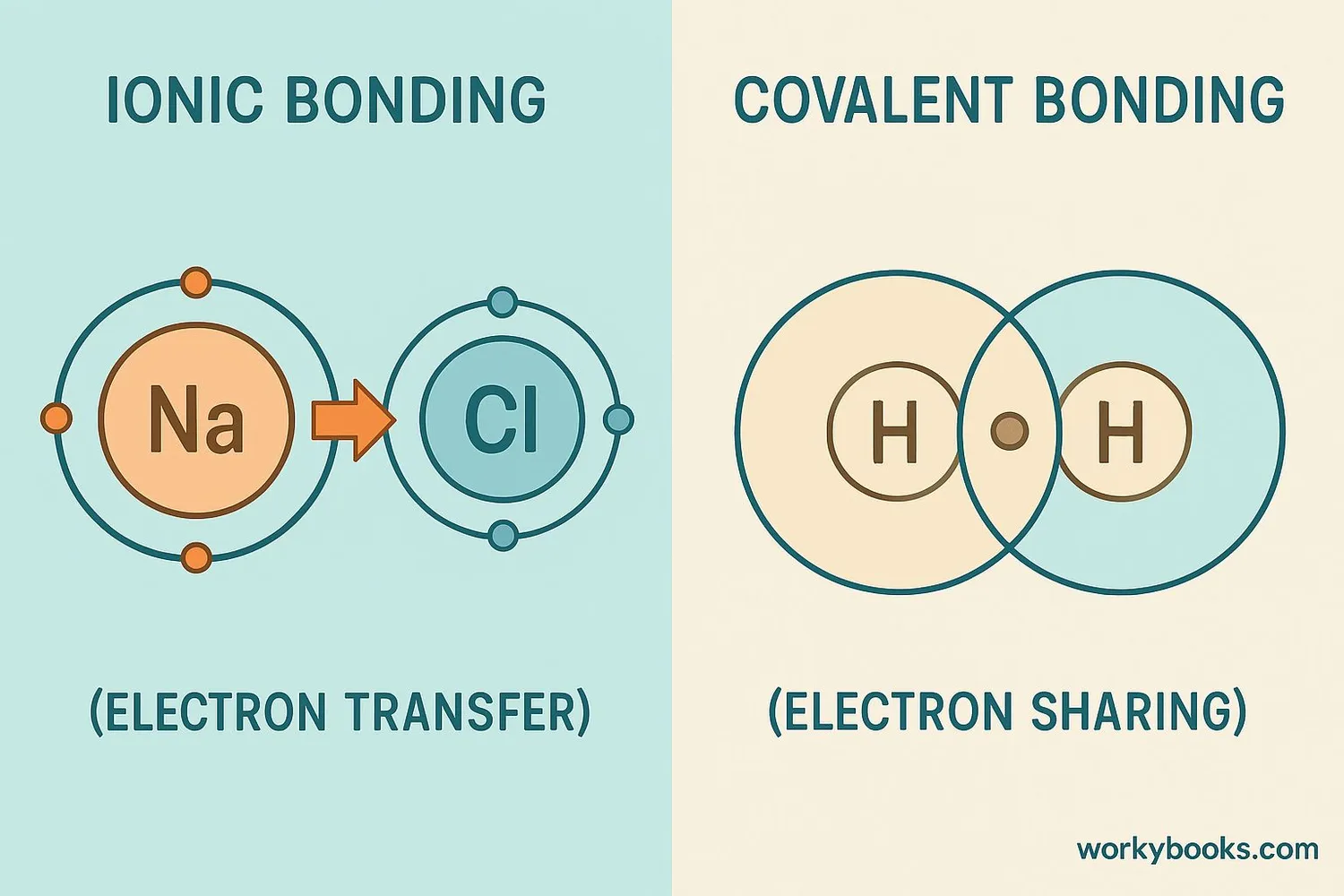
Valence electrons are responsible for chemical bonding! Atoms bond with each other to achieve a stable electron configuration, usually with 8 valence electrons (octet rule).
Ionic Bonds
Atoms transfer electrons to form positive and negative ions that attract each other
Covalent Bonds
Atoms share valence electrons to complete their outer shells
Example: Sodium Chloride (Table Salt)
Sodium (1 valence electron) transfers its electron to Chlorine (7 valence electrons). Sodium becomes Na+ and Chlorine becomes Cl-, and they form an ionic bond.
Example: Water Molecule
Oxygen (6 valence electrons) shares electrons with two Hydrogen atoms (1 valence electron each) to form covalent bonds.
Electron Dot Diagrams
Lewis dot diagrams show valence electrons as dots around the element symbol. This helps visualize bonding possibilities.
Valence Electrons Quiz
Test your knowledge with this interactive quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to common questions about valence electrons:
Fun Valence Electron Trivia
Discover amazing facts about valence electrons!
Conductivity Champions
Metals are great conductors of electricity because their valence electrons move freely between atoms, creating an "electron sea" that carries electrical current.
Carbon's Special Talent
Carbon has 4 valence electrons, allowing it to form many different types of bonds. This versatility makes carbon the basis of all known life on Earth!
Human Electricity
The electrical signals in your nervous system are created by moving ions (atoms that have gained or lost valence electrons) across cell membranes.
Light Creation
When electrons return to their original energy level after being excited, they release energy as light. This is how neon signs and fireworks create their colors!


