Valence Electrons
Interactive passage with audio narration, comprehension questions, and printable PDF.
2
Present
Open in classroom. No work savedAssign
Track progress per studentQuick play
Share with a link, no accountsDownload PDF
Print-ready, offline practice- Format
- Interactive (Online), Printable (PDF)
- Grades
- 5678
- Subjects
- sciencereadingela
- Standards
- MS-PS1-1
- Languages
- English, Spanish
What's included
Reading passage
Reading comprehension
Audio narration
With word word highlighting
Comprehension quiz
Auto-graded
Writing activity
Open-ended response
Glossary & flashcards
Vocabulary practice
Differentiated version
Adapted for varied levels
Spanish translation
Bilingual support
About this reader
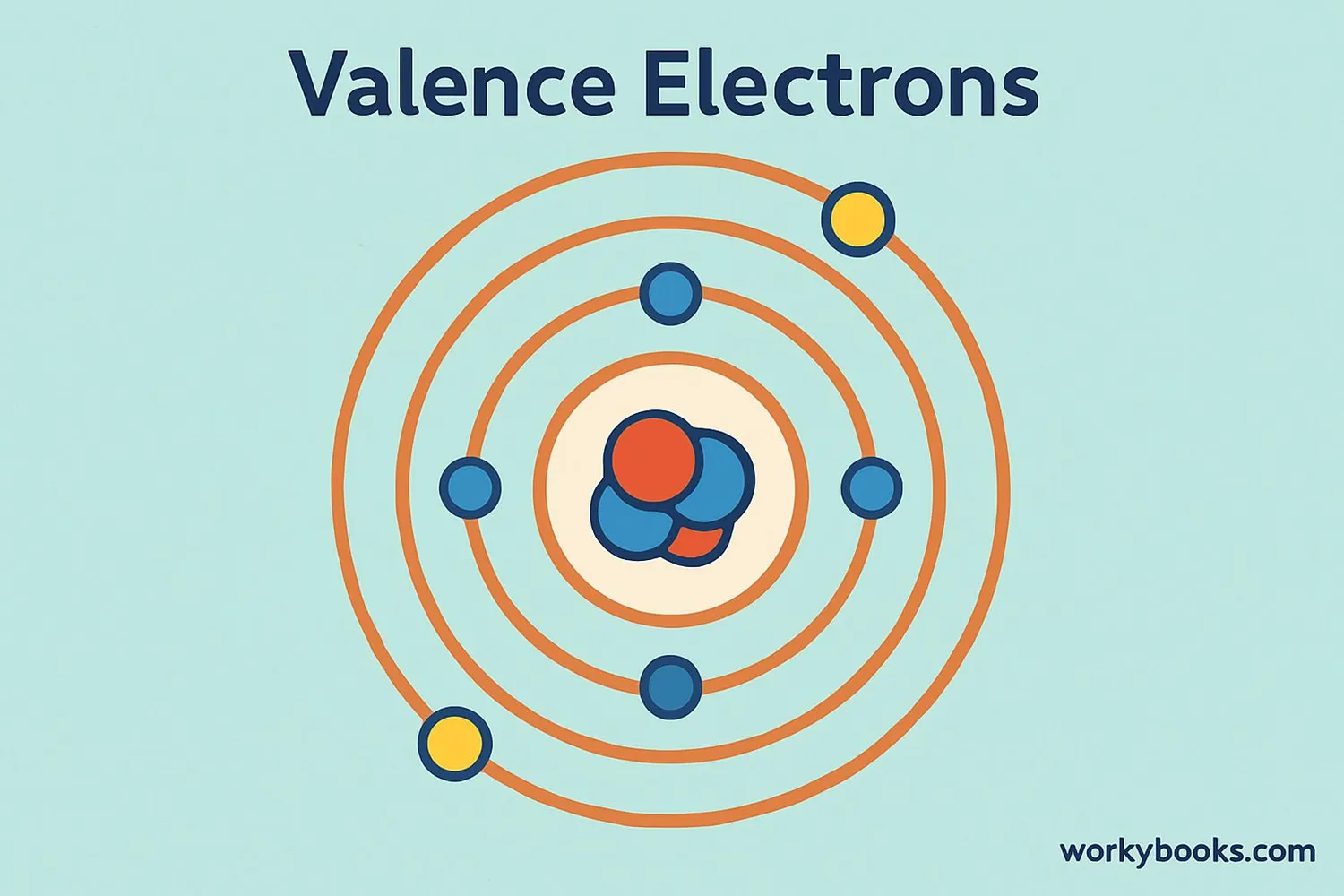
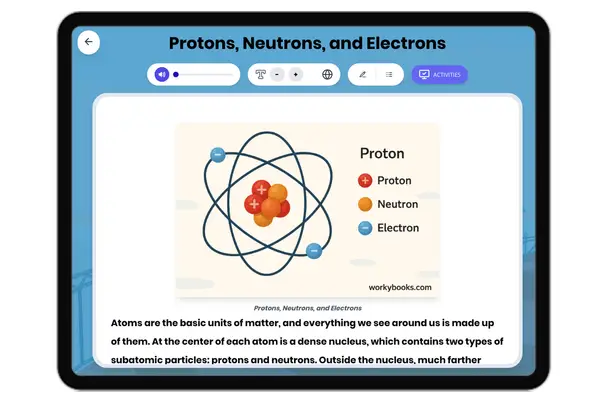
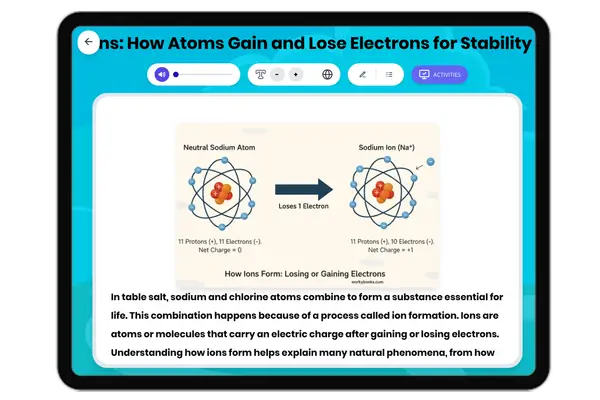
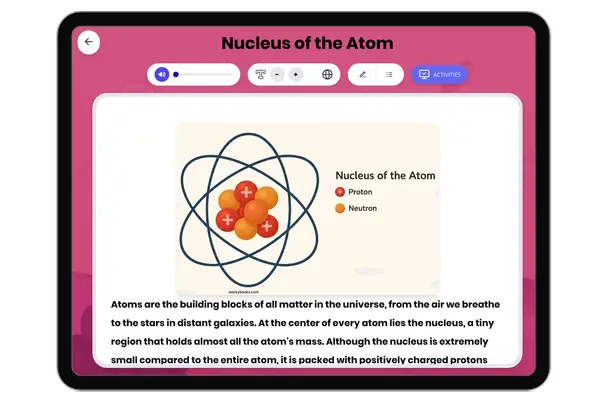
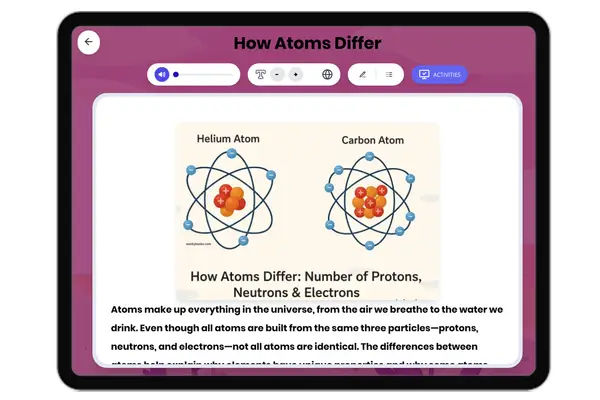
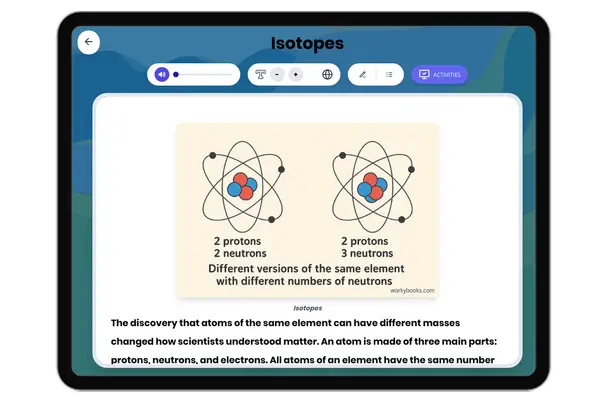
This middle school science passage introduces students to the concept of valence electrons, the outermost electrons that control how atoms interact and combine. Aligned with NGSS MS-PS1-1, it explains how the number of valence electrons can be determined by an element’s group on the periodic table, and how this affects chemical stability and bonding. Through clear explanations and real-world examples like sodium, oxygen, and neon, students explore how valence electrons drive the formation of molecules and influence the behavior of different elements. The passage integrates academic vocabulary, relevant context, and connects to broader scientific principles of matter and chemical processes. Designed for grades 6-8, it also includes a simplified version for differentiated learning, an audio component for accessibility, and interactive activities such as multiple-choice questions, writing prompts, and graphic organizers. This comprehensive resource deepens conceptual understanding of atomic structure, chemical reactivity, and the periodic table.
Written by Workybooks TeamPublished by Workybooks
Preview
Sample passage and quiz content
CONTENT PREVIEW
Who it's for
Perfect for the way you teach
Teachers
- Build comprehension skills
- Auto-graded quiz
- Differentiated reading
Parents
- Read together at home
- Improve fluency
- Quiet reading time
Homeschoolers
- Reading curriculum support
- Independent practice
- Track Lexile growth
Topics
valence electronsperiodic tablechemical bondingstabilityatomsmiddle school scienceNGSS MS-PS1-1