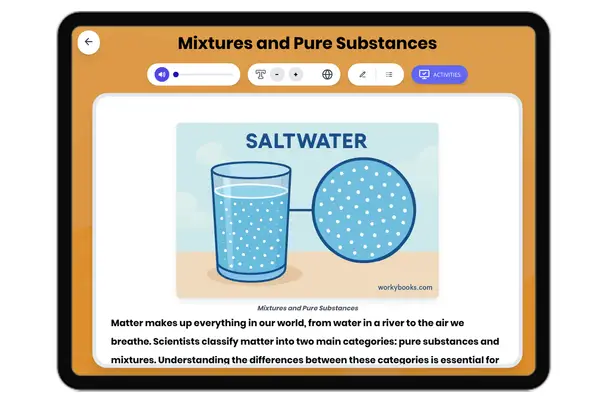
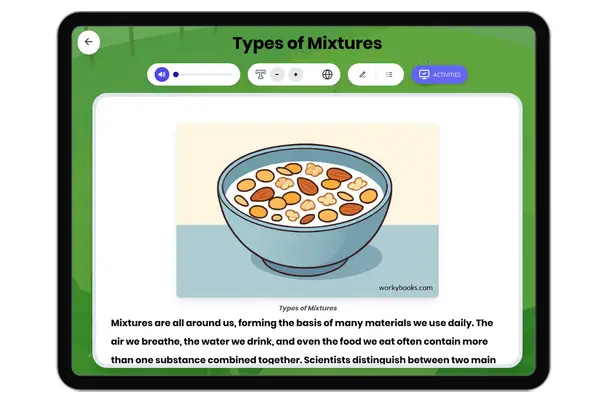
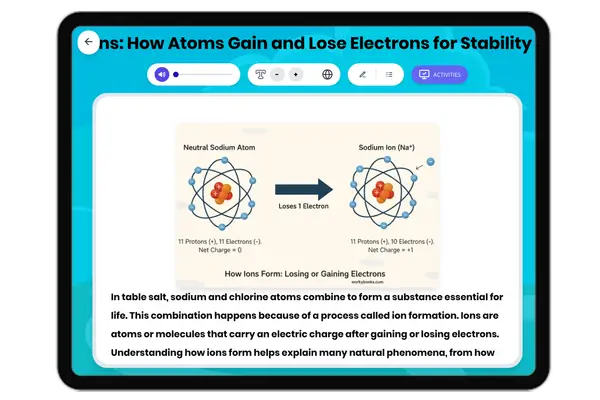
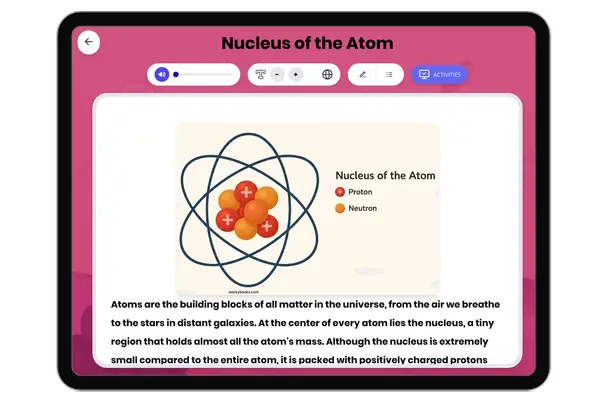
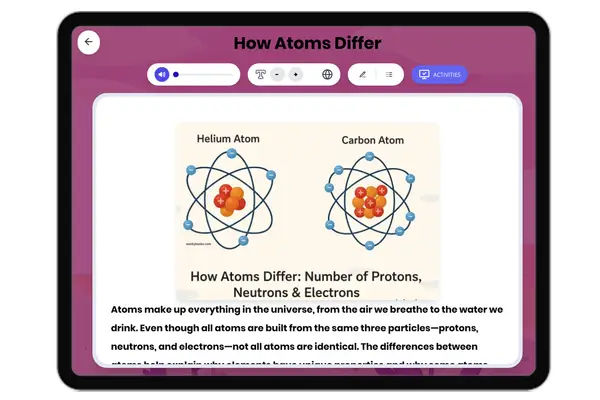
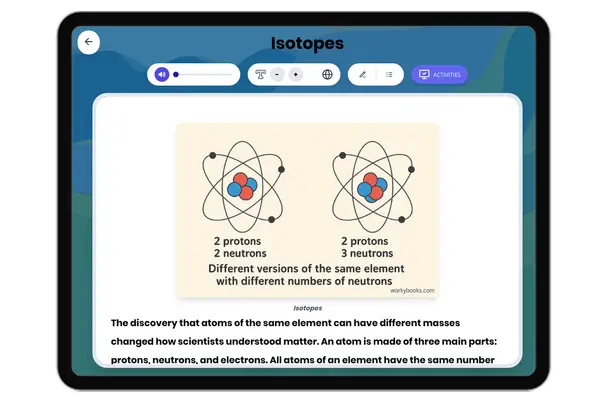
Atomic Structure
Interactive passage with audio narration, comprehension questions, and printable PDF.
510
Present
Open in classroom. No work savedAssign
Track progress per studentQuick play
Share with a link, no accountsDownload PDF
Print-ready, offline practice- Format
- Interactive (Online), Printable (PDF)
- Grades
- 5678
- Subjects
- elareadingscience
- Standards
- MS-PS1-1RI.6.3RI.7.1
- Languages
- English, Spanish
What's included
Reading passage
Reading comprehension
Audio narration
With word word highlighting
Comprehension quiz
Auto-graded
Writing activity
Open-ended response
Glossary & flashcards
Vocabulary practice
Differentiated version
Adapted for varied levels
Spanish translation
Bilingual support
About this reader
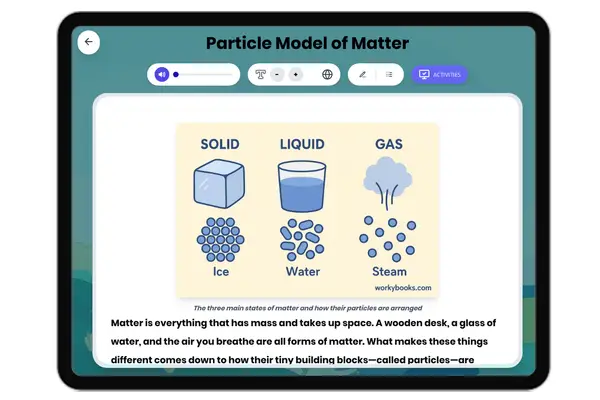
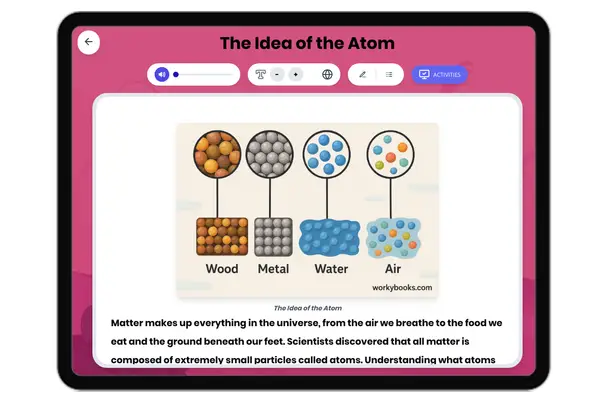
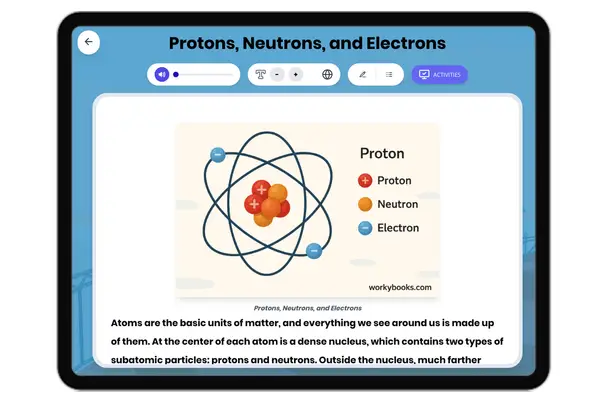
This reading passage on Atomic Structure provides middle school students with an accessible explanation of the fundamental building blocks of matter. The content aligns with NGSS standard MS-PS1-1 (Develop models to describe the atomic composition of simple molecules and extended structures) and Common Core Reading Standards for Informational Text (Key Ideas and Details). The passage is structured with clear subheadings that guide students through key concepts: what atoms are, the structure of the nucleus, the behavior of electrons, how atoms determine elements, molecule formation, and scientific tools for studying atoms. With a Flesch reading score of 65, this 229-word passage uses simple language and familiar examples like water molecules to make abstract concepts concrete for grades 6-8 students. This resource sits within the Science→Physical Science→Properties of Matter domain and helps students visualize the invisible structures that make up everything in our world.
Written by Workybooks TeamPublished by Workybooks
Preview
Sample passage and quiz content
CONTENT PREVIEW
Who it's for
Perfect for the way you teach
Teachers
- Build comprehension skills
- Auto-graded quiz
- Differentiated reading
Parents
- Read together at home
- Improve fluency
- Quiet reading time
Homeschoolers
- Reading curriculum support
- Independent practice
- Track Lexile growth
Topics
atomic structuremiddle school scienceprotonsneutronselectronsnucleuselementsmoleculesatomsparticle charges