Acetic Acid - Definition, Examples, Quiz, FAQ, Trivia
Discover the science behind vinegar and how this special chemical affects our world!
What is Acetic Acid?
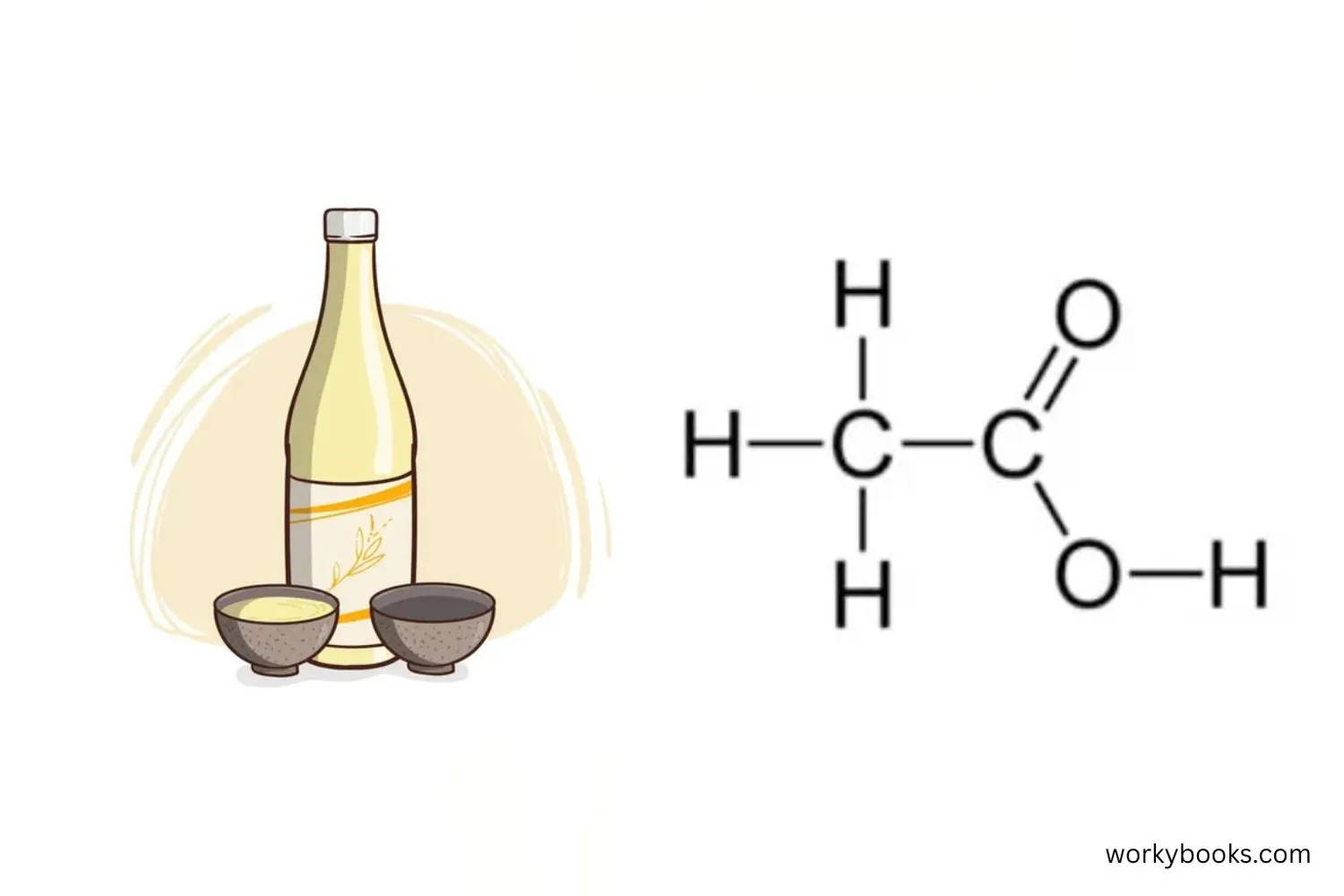
Acetic acid is an organic acid that gives vinegar its sour taste and strong smell. It's a type of carboxylic acid found in many foods and products we use every day.
Here are some key facts about acetic acid:
- Chemical formula: CH₃COOH (that's 2 carbon, 4 hydrogen, and 2 oxygen atoms)
- Found naturally in vinegar (about 5-8% concentration)
- Classified as a weak acid because it doesn't completely break apart in water
- One of the simplest carboxylic acids
- Has been used by humans for thousands of years as a food preservative
Science Fact!
The word "acetic" comes from the Latin word "acetum" meaning vinegar. Humans have used vinegar made from acetic acid for over 10,000 years!
Properties & Structure
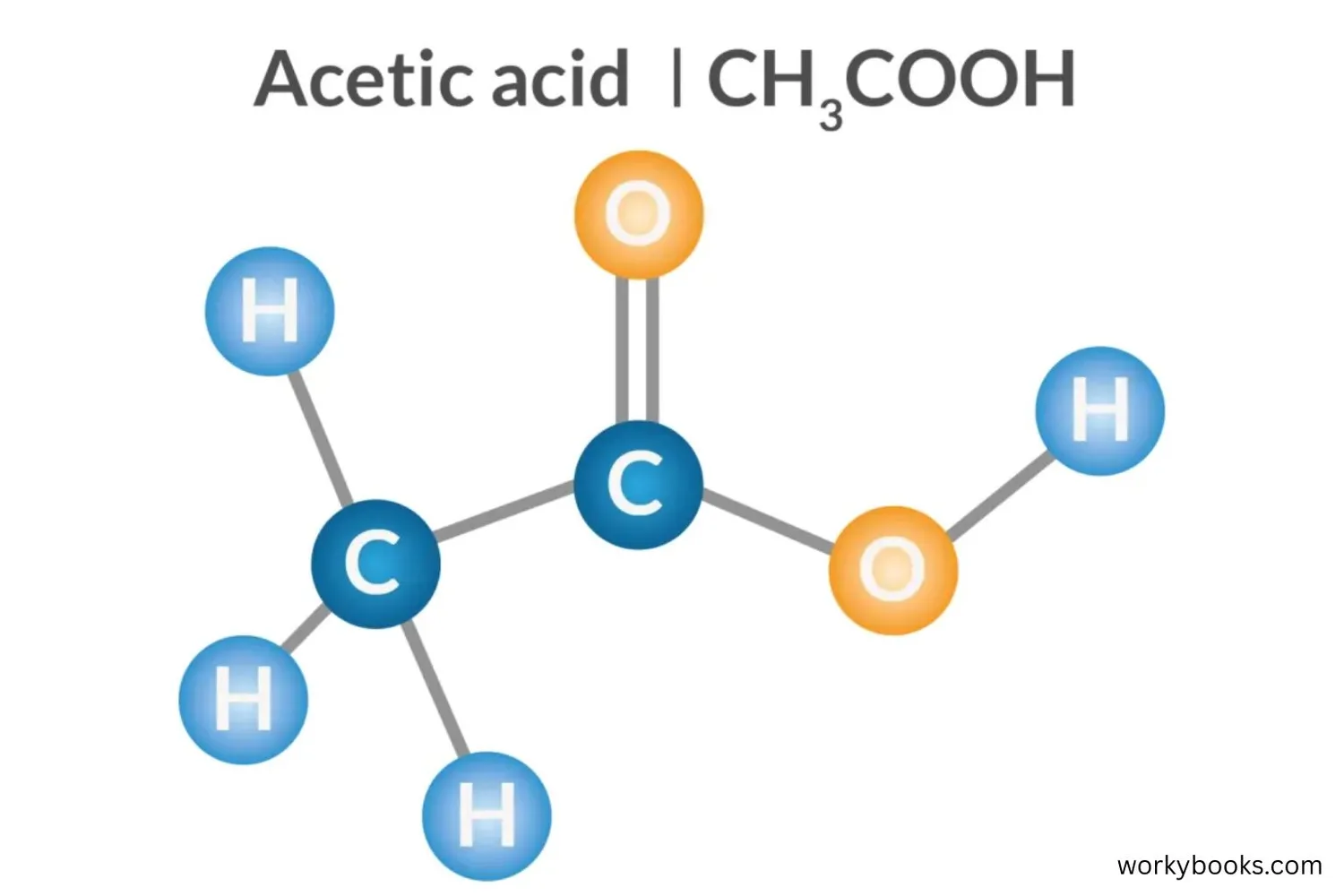
Acetic acid has special properties that make it useful in many ways. Let's explore its physical and chemical characteristics:
Physical Properties
Clear, colorless liquid with a strong, pungent vinegar smell
Taste
Sour taste that you recognize from vinegar
Solubility
Mixes completely with water, alcohol, and other solvents
Acidity Level
Weak acid with pH around 2.4 when undiluted
Chemical Structure
Contains a carboxylic acid group (-COOH) that makes it acidic
The special structure of acetic acid gives it its properties. The carboxylic acid group (-COOH) can donate a hydrogen ion (H⁺), which makes it an acid. This is why vinegar can dissolve minerals and react with baking soda!
Chemistry Fact!
Pure acetic acid is sometimes called "glacial acetic acid" because it freezes just below room temperature at 62°F (17°C), forming ice-like crystals!
How Acetic Acid is Made
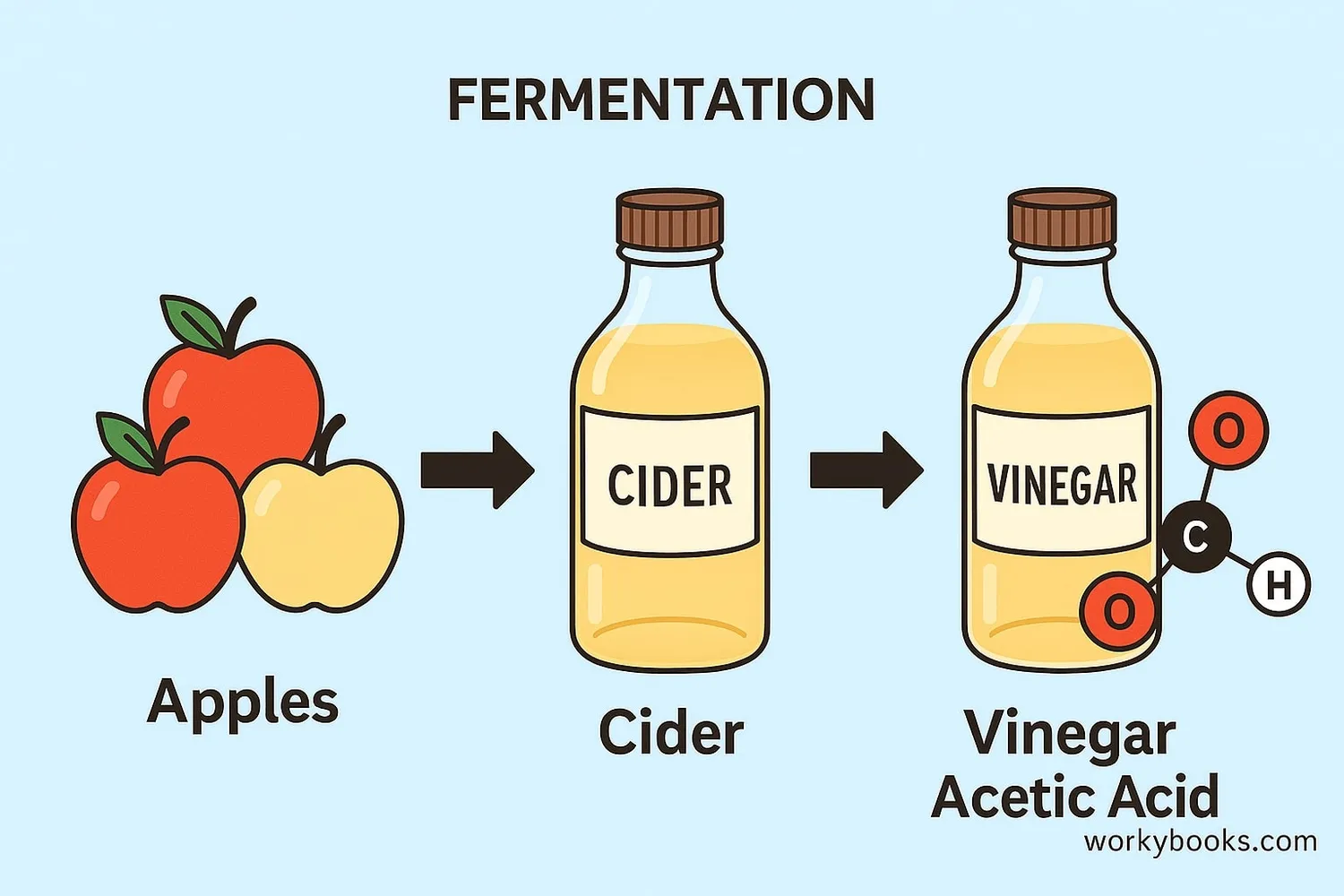
Acetic acid can be produced in different ways. The two main methods are:
Natural Fermentation
Bacteria convert alcohol into acetic acid (how vinegar is made)
Industrial Production
Chemical process combining methanol and carbon monoxide
Fermentation Process:
This natural method uses Acetobacter bacteria to convert ethanol (alcohol) into acetic acid. Here's how it works:
1. Fruits or grains are fermented to produce alcohol (like wine or cider)
2. Acetobacter bacteria are added
3. Bacteria convert alcohol to acetic acid using oxygen from air
4. After several weeks or months, vinegar is formed
Industrial Method:
Most acetic acid used in industry is made through a chemical process called methanol carbonylation. This method combines methanol and carbon monoxide using special catalysts to produce acetic acid more quickly and in larger quantities.
Uses of Acetic Acid
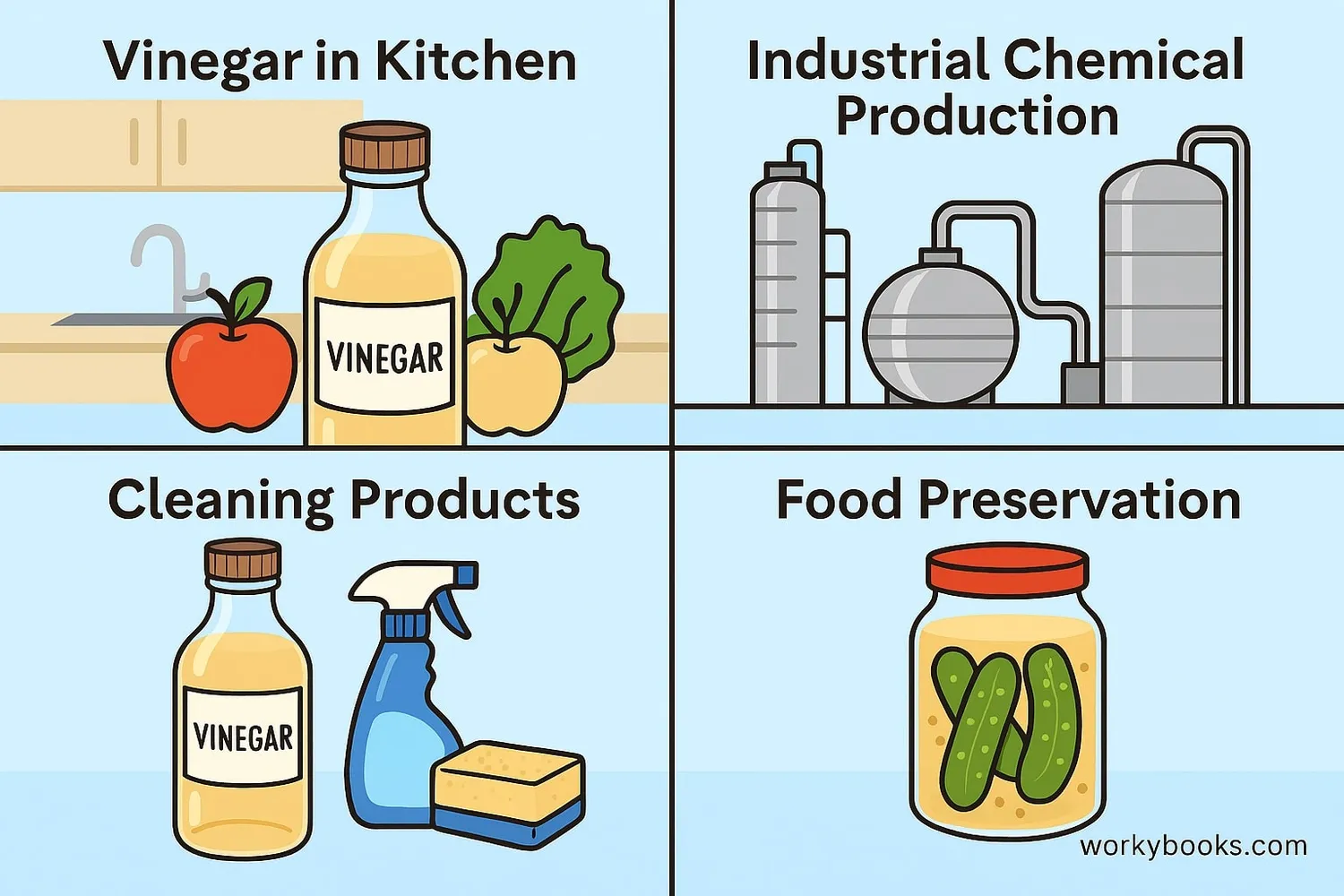
Acetic acid has many important uses in our daily lives and in industry:
Cooking & Food
Vinegar for cooking, pickling, and salad dressings
Cleaning
Natural cleaner for surfaces, windows, and appliances
Industry
Production of plastics, paints, and synthetic fabrics
Other important uses include:
• Food preservative: Prevents growth of harmful bacteria in pickled foods
• Laboratory chemical: Used in science experiments and testing
• Medicine: Some ear infections are treated with acetic acid solutions
• Agriculture: Used as a natural herbicide to control weeds
The reason acetic acid has so many uses is because it's a versatile chemical that can dissolve many substances, kill microorganisms, and participate in chemical reactions to create new materials.
Acetic Acid Knowledge Quiz
Test what you've learned about acetic acid with this quiz! Answer all 5 questions to see how much you know.
Frequently Asked Questions
Here are answers to common questions about acetic acid:
Acetic Acid Trivia
Discover some amazing facts about acetic acid!
Ancient Cleaning
Roman soldiers used vinegar as a refreshing drink and to clean their equipment! They believed it gave them strength and prevented illness.
Massive Production
Over 15 billion pounds of acetic acid are produced worldwide each year! That's enough to fill more than 1,000 Olympic-sized swimming pools.
Special Property
Pure acetic acid is called "glacial" because it freezes at 62°F (17°C), forming ice-like crystals at slightly below room temperature.
Food Preservation
Vinegar's acidity prevents harmful bacteria growth. This is why pickled foods can last for months without refrigeration!


