Electrolysis - Definition, Examples, Quiz, FAQ, Trivia
Discover how electricity transforms chemicals into new substances!
What is Electrolysis?
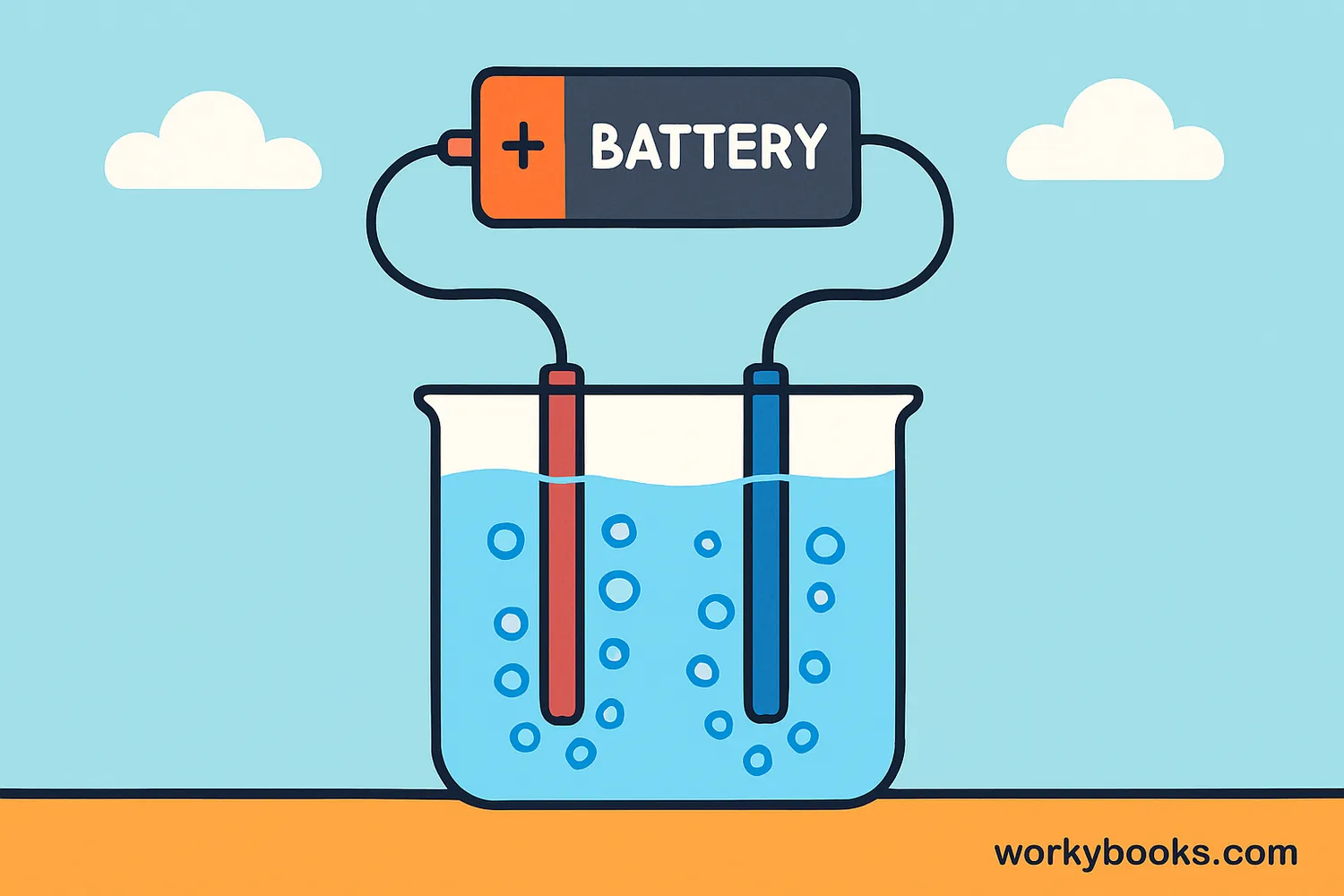
Electrolysis is a fascinating process where we use electricity to break down chemical compounds into simpler substances. It's like using electrical energy to perform chemistry magic!
During electrolysis, an electric current passes through a liquid called an electrolyte, causing chemical reactions that split compounds into their basic elements. For example, electricity can split water into hydrogen and oxygen gases.
Key Terms
Electrolyte: A substance that conducts electricity when dissolved or molten
Electrodes: Conductors through which electricity enters or leaves the electrolyte
How Electrolysis Works
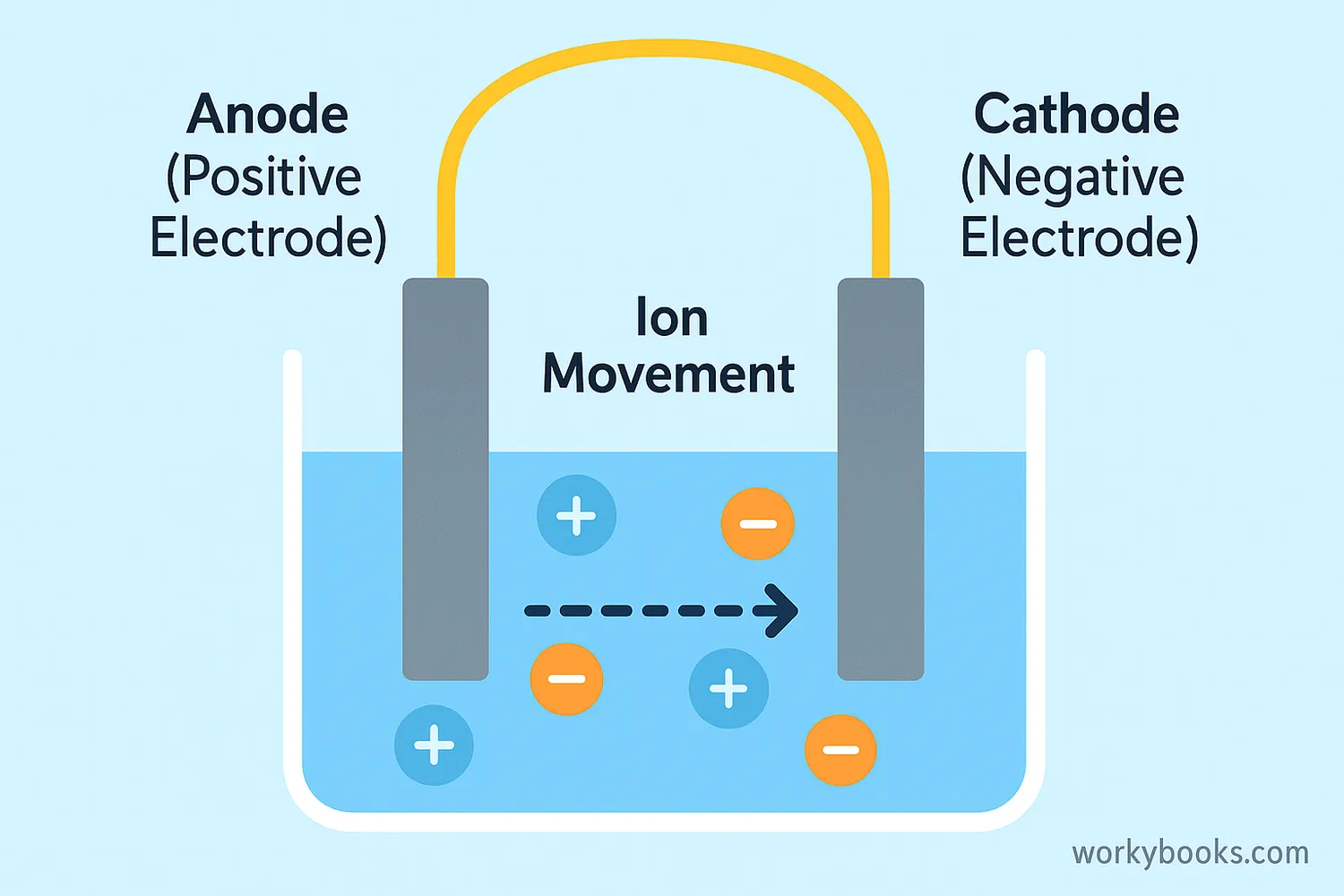
Electrolysis requires three main components:
1. A power source (like a battery) that provides electrical energy
2. Two electrodes that conduct electricity
3. An electrolyte that contains ions
Anode (+)
Oxidation occurs here - ions lose electrons
Cathode (-)
Reduction occurs here - ions gain electrons
Electrolyte
Contains ions that carry electrical current
During electrolysis of water:
• At the cathode: 2H₂O + 2e⁻ → H₂ + 2OH⁻ (hydrogen gas forms)
• At the anode: 2H₂O → O₂ + 4H⁺ + 4e⁻ (oxygen gas forms)
Types of Electrolysis
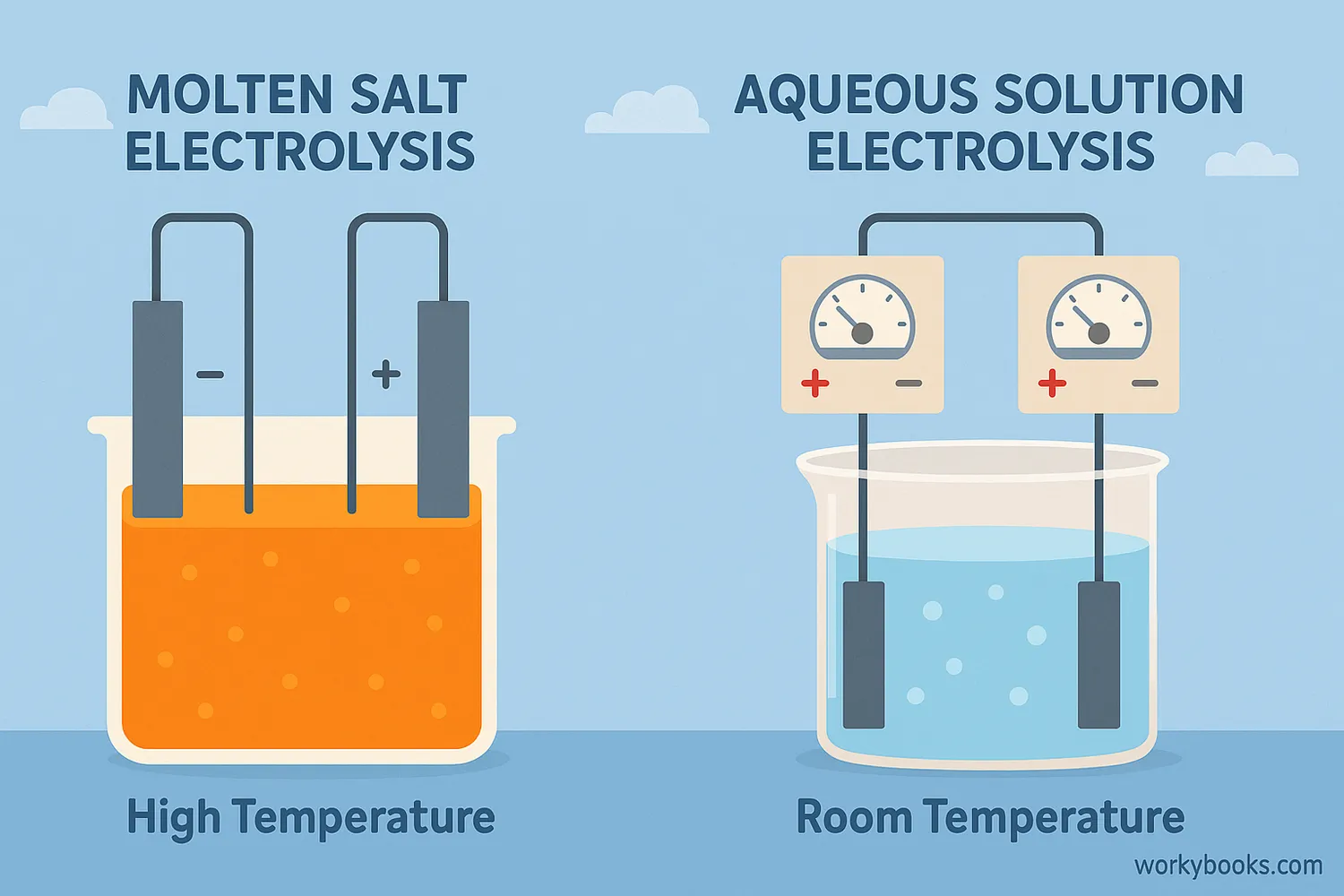
There are two main types of electrolysis:
Molten Salt Electrolysis
Used for compounds with very high melting points. The solid is heated until it melts, allowing ions to move freely.
Aqueous Solution Electrolysis
The compound is dissolved in water. This method is used for substances that dissolve easily.
Faraday's Laws
Michael Faraday discovered two important laws about electrolysis:
1. The amount of chemical change is proportional to the charge passed
2. The masses of different substances produced are proportional to their equivalent weights
Real-World Uses
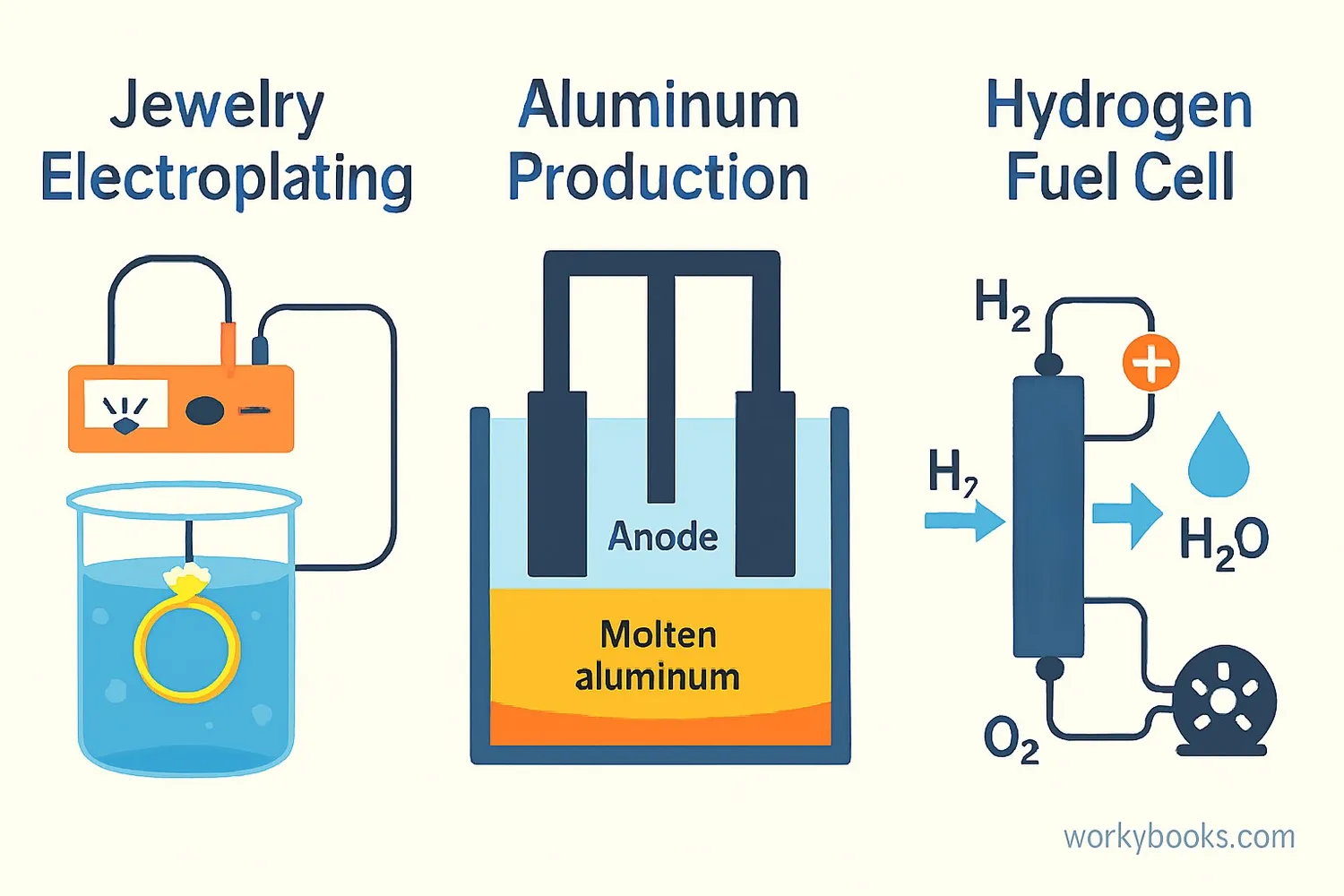
Electrolysis has many important applications in our daily lives and industries:
Electroplating
Coating objects with a thin layer of metal to make them shiny or prevent corrosion
Hydrogen Production
Creating hydrogen fuel for clean energy vehicles
Metal Extraction
Obtaining pure metals like aluminum from their ores
Other applications include:
• Purifying copper for electrical wiring
• Producing chlorine for water treatment
• Manufacturing sodium hydroxide (lye) for soaps
• Creating pure substances for medicines
Electrolysis Quiz
Test your knowledge with this electrolysis quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to common questions about electrolysis:
Electrolysis Trivia
Discover some amazing facts about electrolysis!
Historical Discovery
The term "electrolysis" was introduced by Michael Faraday in the 1830s. He also coined words like electrode, electrolyte, anode, and cathode that we still use today!
Space Applications
Electrolysis is used on the International Space Station to split water into oxygen for astronauts to breathe and hydrogen for fuel cells.
Massive Production
Over 70 million tons of aluminum are produced worldwide each year using electrolysis. That's equivalent to about 7 million school buses!
Electric Vehicles
Hydrogen fuel for clean cars is produced through electrolysis. When burned, it only produces water, making it a pollution-free fuel source.


