Activation Energy - Definition, Examples, Quiz, FAQ, Trivia
Discover how chemical reactions get started with the energy boost they need!
What is Activation Energy?
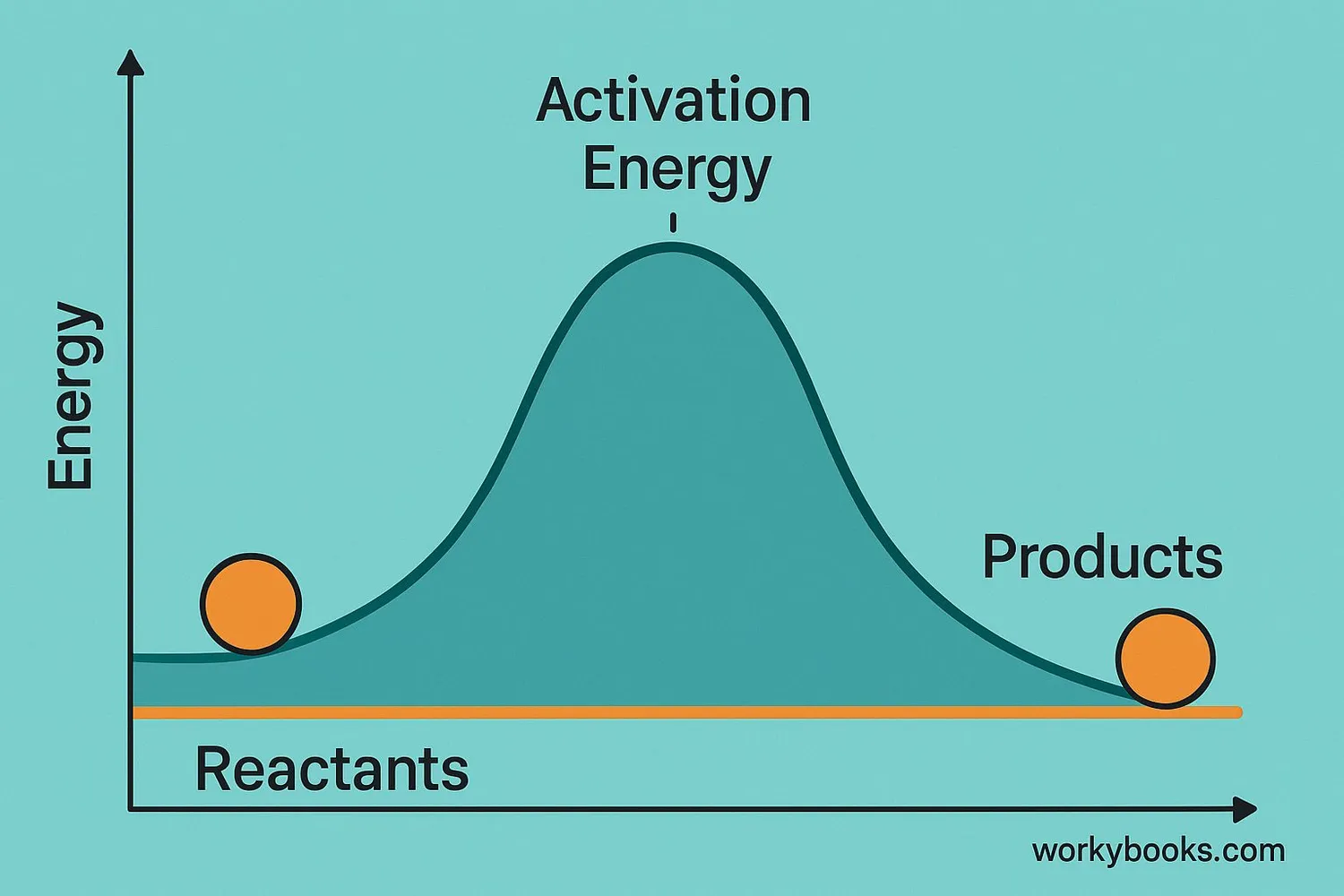
Activation energy is the minimum amount of energy needed to start a chemical reaction. Think of it like the push you need to get a ball rolling over a hill - once it's over the top, it rolls down easily!
Every chemical reaction needs this initial energy boost to get started. It's why some reactions need heat, light, or electricity to begin. Once started, many reactions release energy to keep going.
Reactants
Starting materials for the reaction
Activation Energy
Energy needed to start the reaction
Transition State
High-energy point of the reaction
Products
The new substances formed
Energy Fact!
Even reactions that release energy overall need activation energy to get started, like a match needs to be struck to light a fire.
Energy Barriers in Reactions
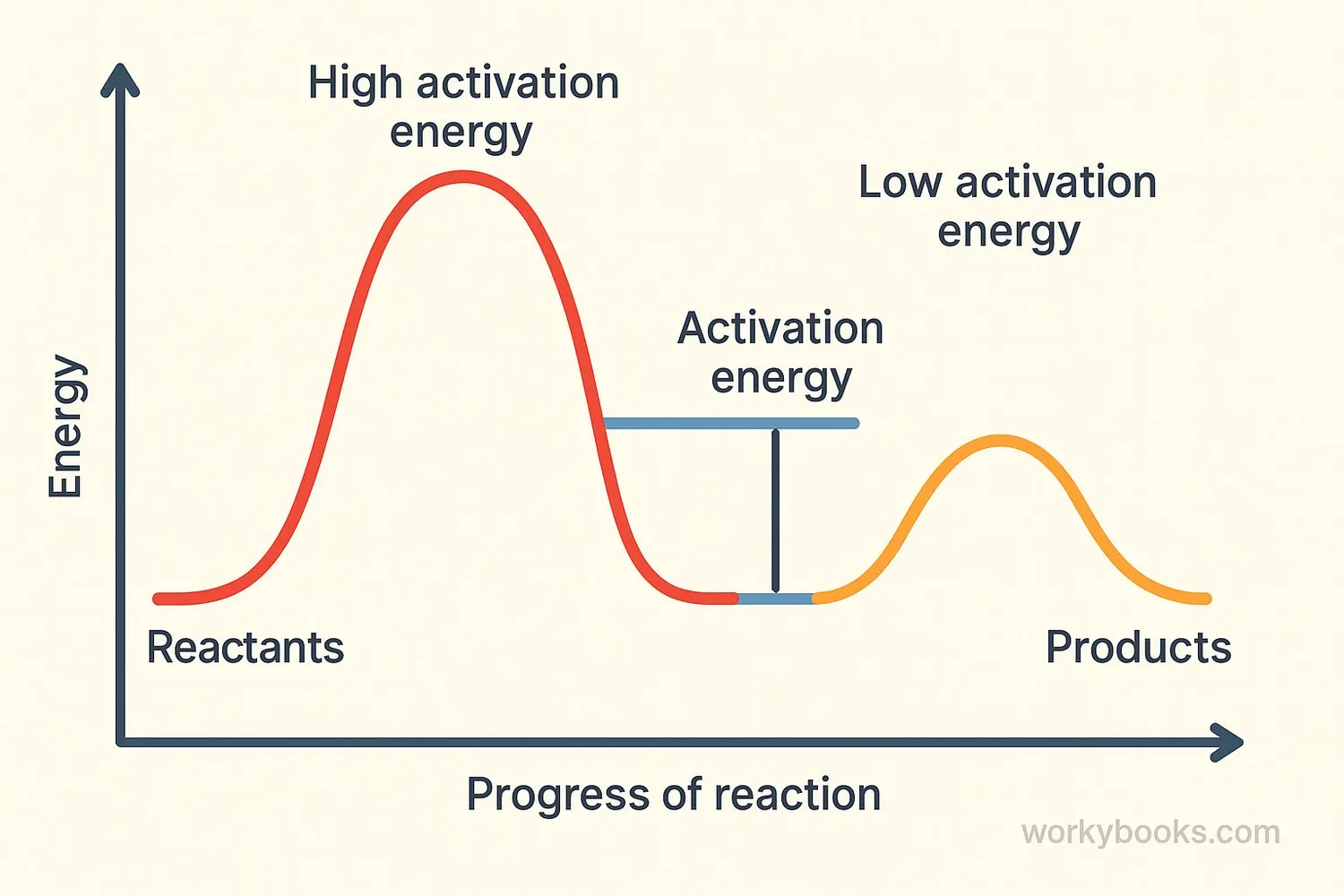
Chemical reactions face an energy barrier that they must overcome. This barrier determines how easily and quickly a reaction can happen:
High activation energy = Harder to start, slower reaction
Low activation energy = Easier to start, faster reaction
The height of this energy barrier depends on the specific chemicals involved and the type of reaction. Some reactions need just a little energy to start, while others need a lot!
Reaction Kinetics
Reaction kinetics studies how different conditions like temperature, concentration, and catalysts affect reaction rates by changing the activation energy needed.
Catalysts Lower Activation Energy
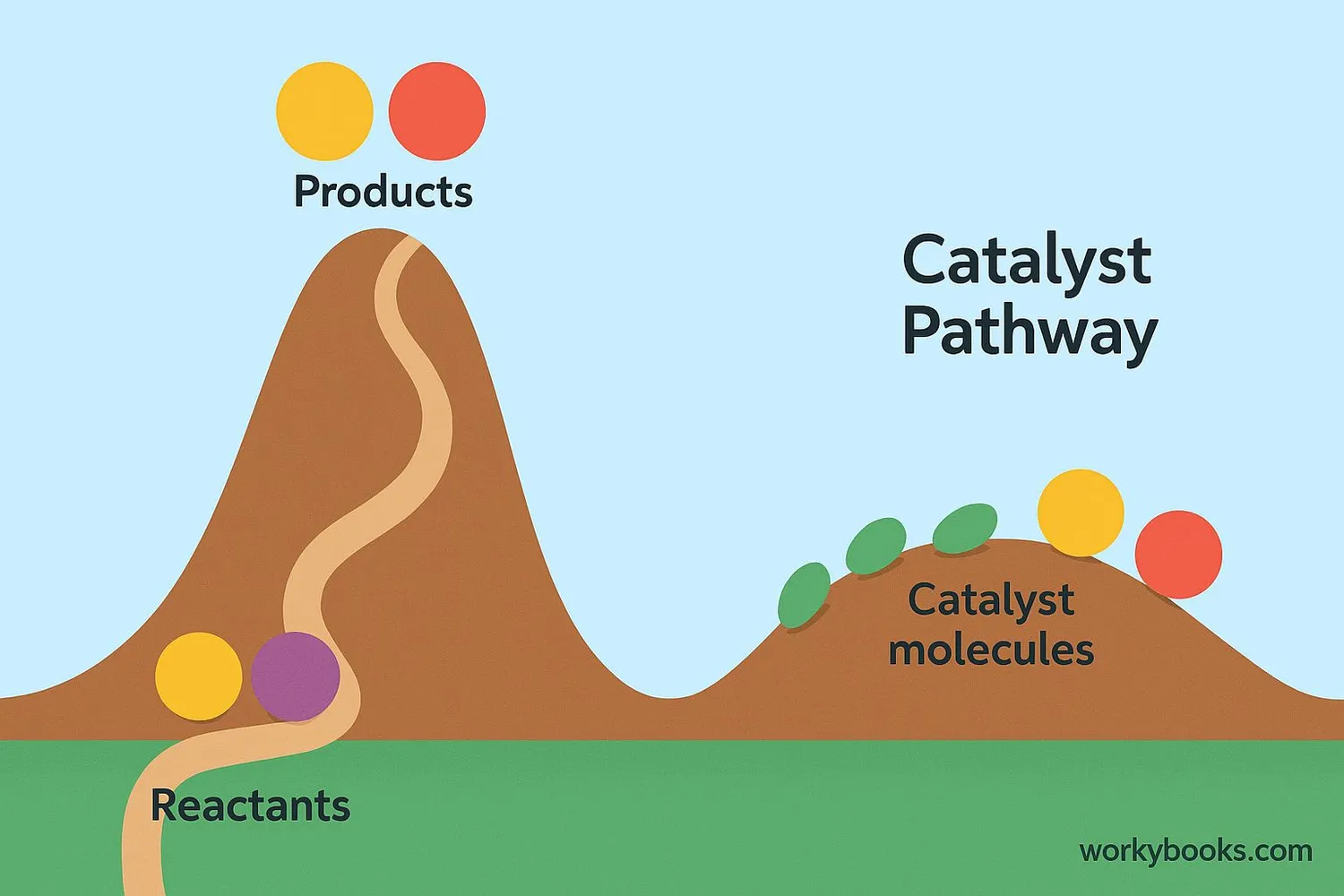
A catalyst is a special helper that speeds up chemical reactions without being used up. Catalysts work by providing an alternative pathway for the reaction that has lower activation energy.
Enzymes are biological catalysts in living things that help important reactions happen quickly at body temperature. Without enzymes, many reactions in your body would be too slow to sustain life!
Alternative Pathway
Catalysts create a different reaction route with lower energy requirements
Reusable
Catalysts aren't consumed and can be used repeatedly
Lower Temperatures
Reactions can happen faster at lower temperatures
Enzyme Function
Your body contains thousands of enzymes! For example, amylase in saliva breaks down starch, and catalase breaks down hydrogen peroxide in cells.
Activation Energy in Everyday Life

Activation energy is all around us! Here are some common examples:
Starting a Fire
The match provides activation energy to begin burning
Car Engines
The spark plug provides activation energy to ignite fuel
Cooking Food
Heat provides activation energy for chemical changes
In living things, enzymes act as catalysts to lower activation energy for essential reactions:
• Digestion: Enzymes break down food molecules
• Respiration: Enzymes help release energy from glucose
• DNA replication: Enzymes copy genetic information
Activation Energy Quiz
Test your knowledge about activation energy with this quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to some common questions about activation energy:
Fun Activation Energy Trivia
Discover some amazing facts about activation energy!
Speedy Reactions
Some biochemical reactions in your body happen in just 0.0000000001 seconds thanks to enzymes lowering activation energy!
Car Catalysts
Catalytic converters in cars use platinum and palladium to lower activation energy, converting harmful exhaust gases into safer substances.
Cooking Chemistry
When you bake cookies, the oven provides activation energy for the Maillard reaction that creates delicious flavors and golden-brown color.
Enzyme Efficiency
A single enzyme molecule can catalyze up to 1,000,000 reactions per second! Without enzymes, your body couldn't function.


