Decomposition Reactions - Definition, Examples, Quiz, FAQ, Trivia
Discover how complex substances break down into simpler ones in chemical reactions
What is a Decomposition Reaction?
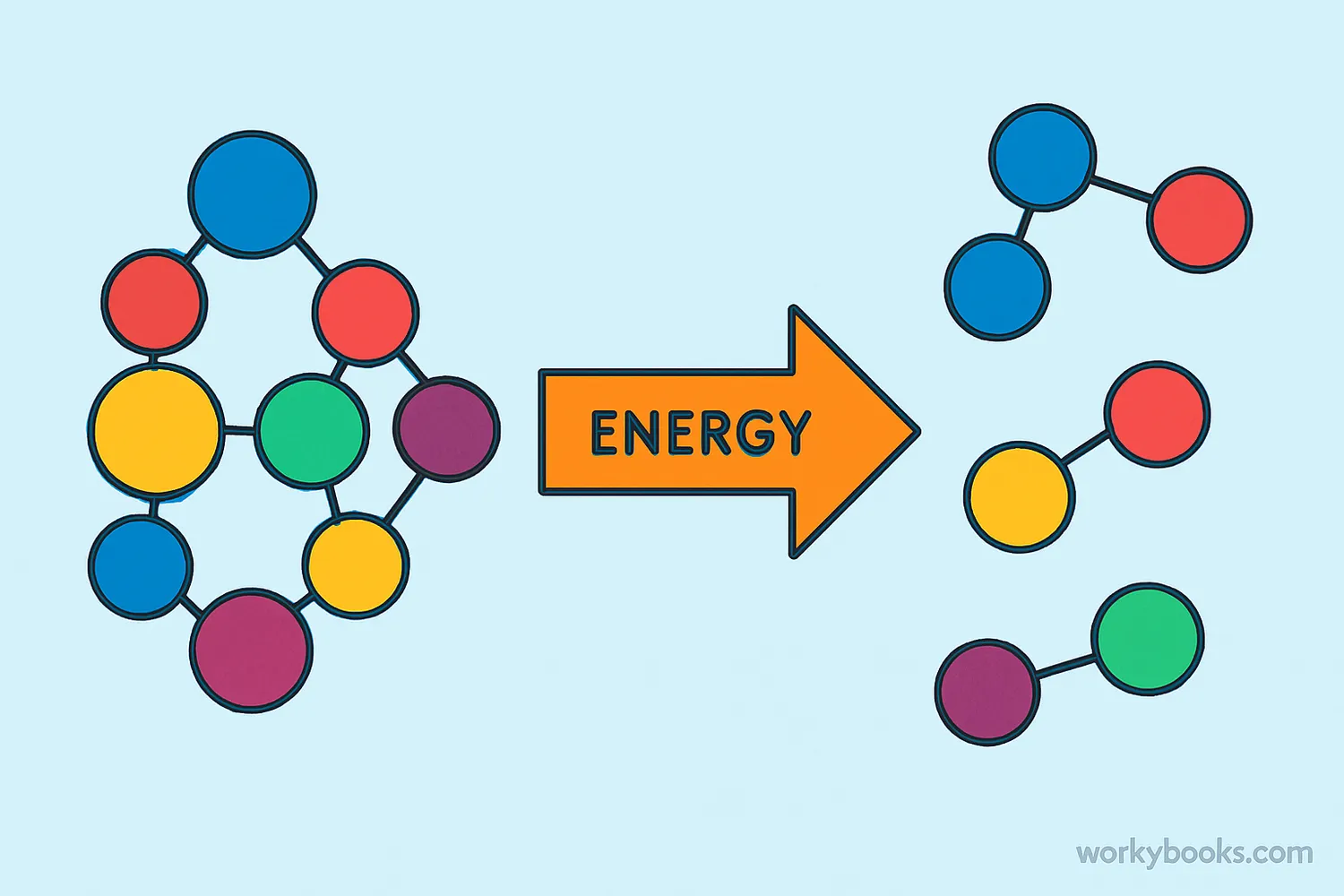
A decomposition reaction is a type of chemical reaction where a single compound breaks down into two or more simpler substances. It's the opposite of a combination reaction. Think of it like breaking a big Lego structure into its individual pieces!
In scientific terms: AB → A + B
For example, when water decomposes, it breaks down into hydrogen and oxygen gases: 2H₂O → 2H₂ + O₂
Key Fact!
Decomposition reactions usually require energy input to get started. This can come from heat, light, or electricity.
Types of Decomposition Reactions
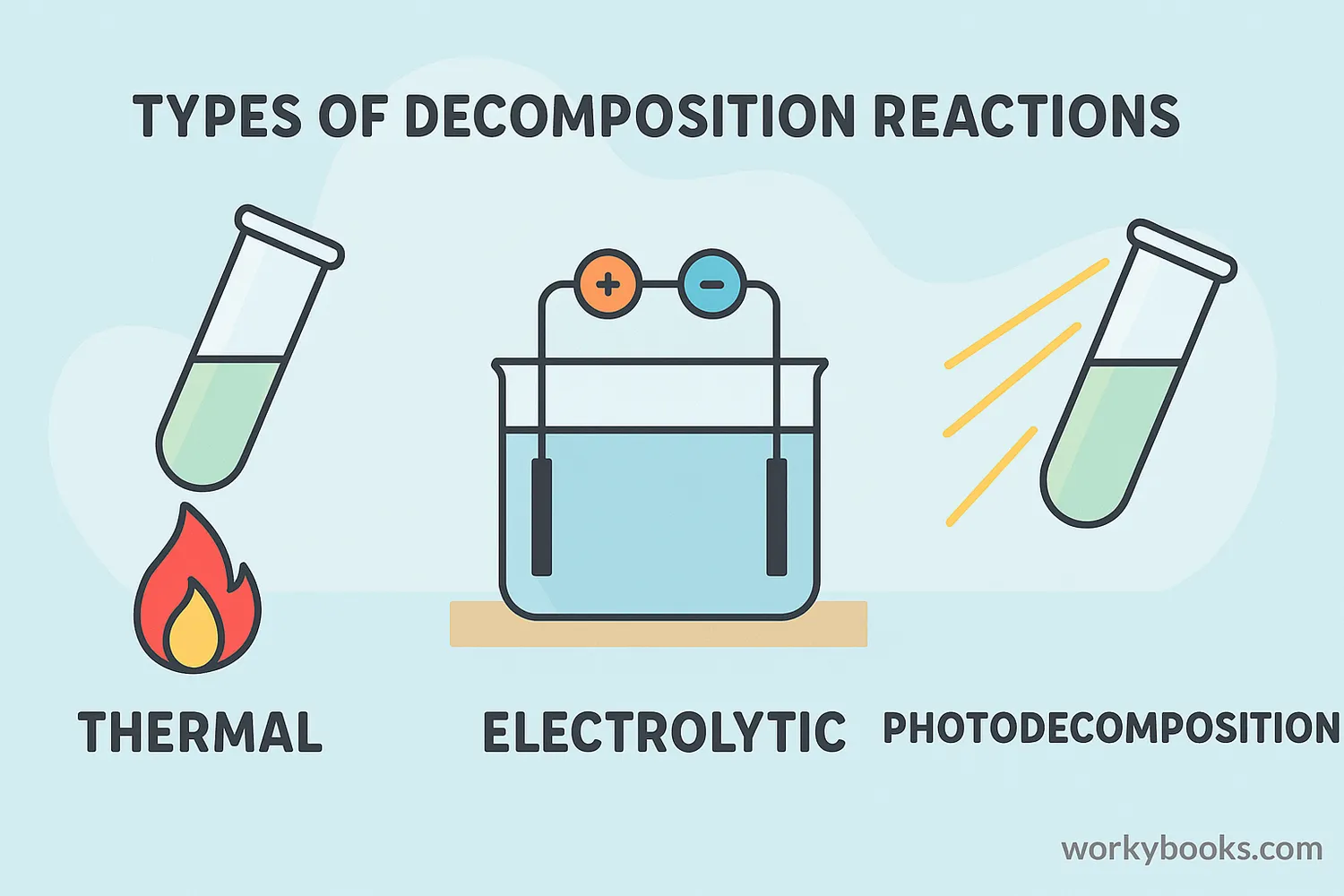
Decomposition reactions are classified by the energy source that causes them to happen:
Thermal Decomposition
Heat causes the breakdown (e.g., baking soda decomposing when heated)
Electrolytic Decomposition
Electricity causes the breakdown (e.g., decomposing water using electricity)
Photodecomposition
Light causes the breakdown (e.g., silver chloride decomposing in sunlight)
2NaHCO₃ (s) → Na₂CO₃ (s) + H₂O (g) + CO₂ (g)
Thermal Decomposition Fact!
When limestone (CaCO₃) is heated, it decomposes into quicklime (CaO) and carbon dioxide. This reaction is used to make cement!
Real-Life Examples
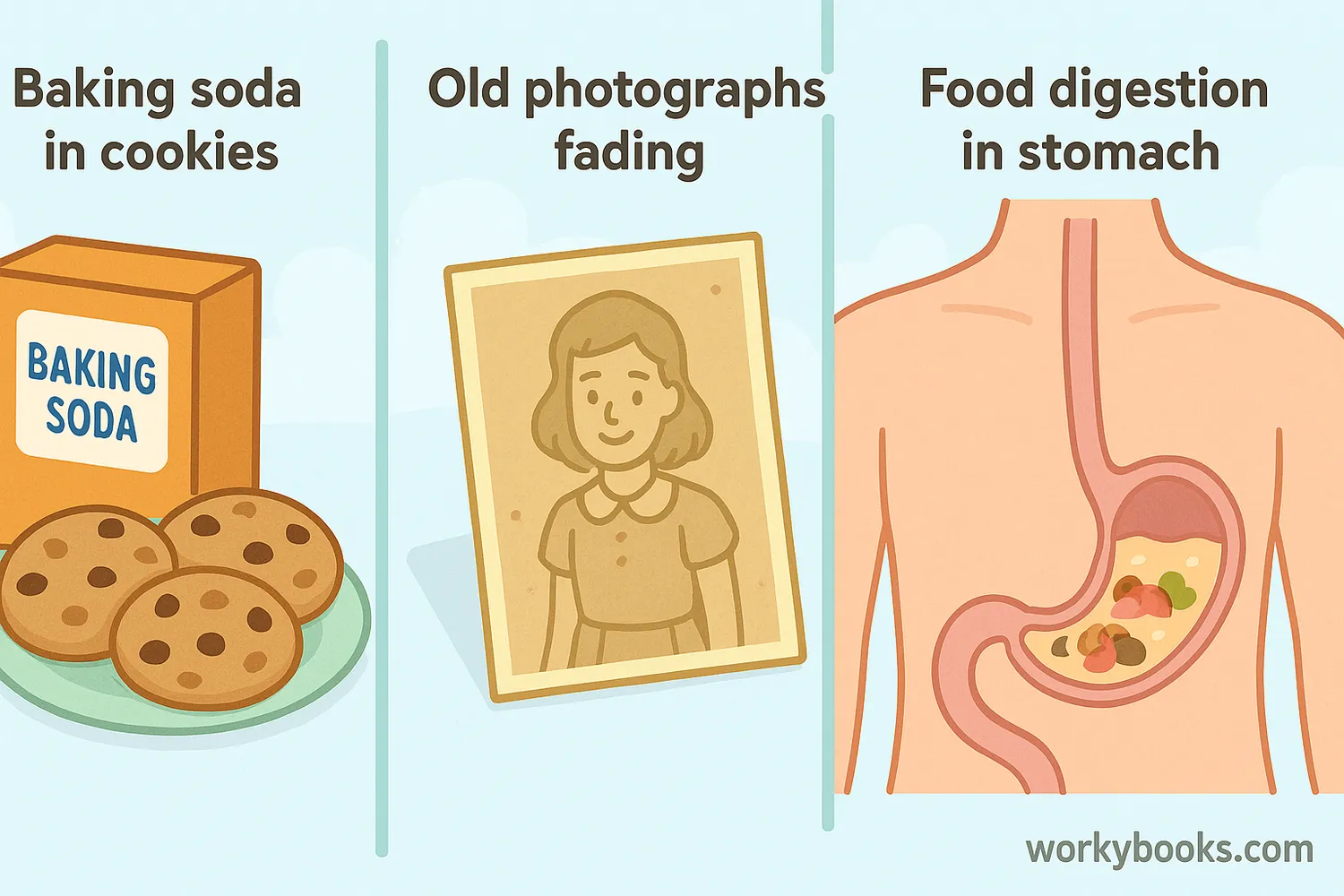
Decomposition reactions happen all around us! Here are some examples you might encounter:
Baking
Baking soda decomposes to produce carbon dioxide gas that makes cakes rise
Photography
Silver compounds decompose when exposed to light, creating photographs
Digestion
Your body decomposes complex food molecules into simpler nutrients
2NaHCO₃ → Na₂CO₃ + H₂O + CO₂
Why Decomposition Reactions Matter
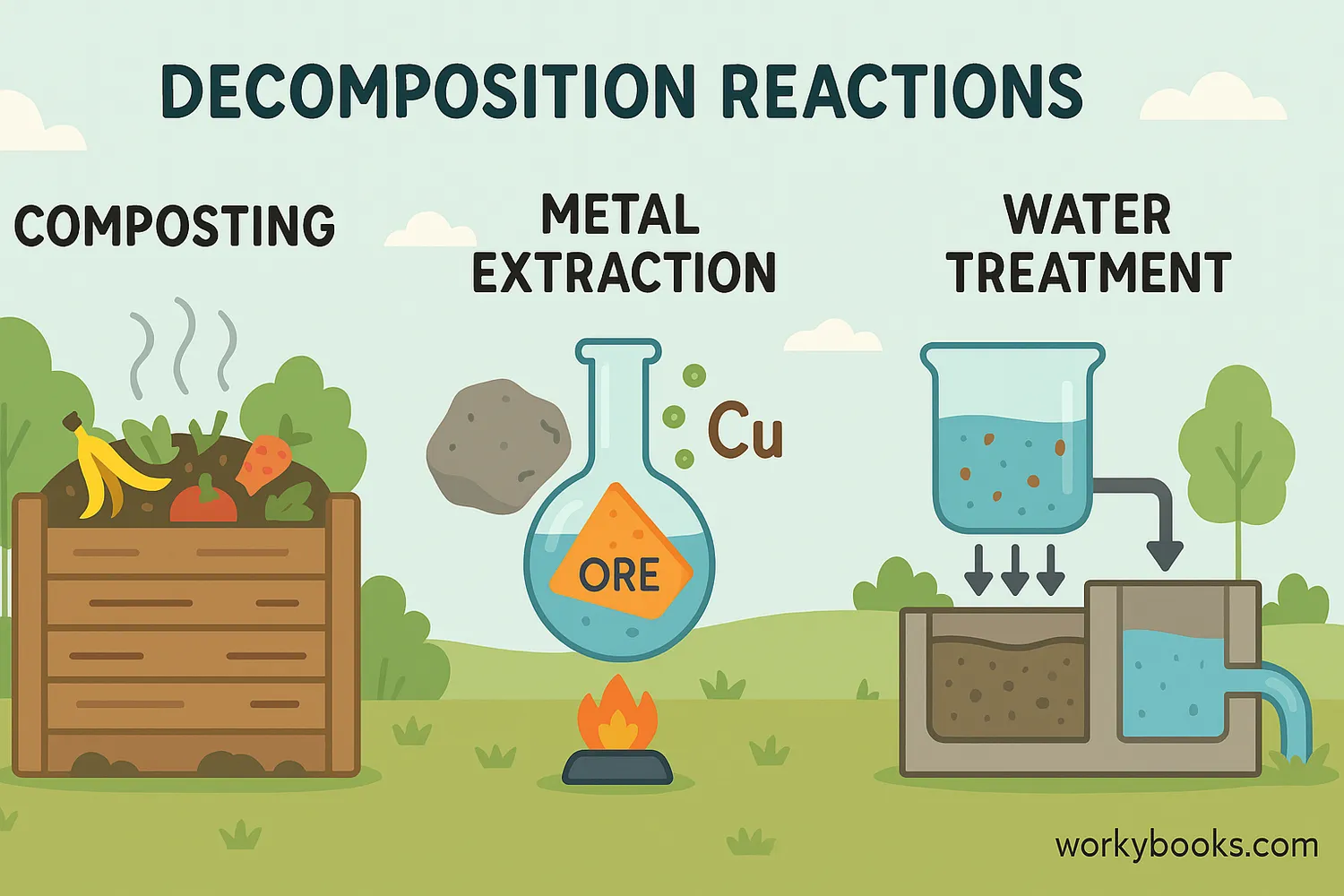
Decomposition reactions play crucial roles in our world:
Nature's Recycling
Decomposition breaks down dead plants and animals, returning nutrients to soil
Industrial Processes
Used to extract metals from ores and produce important chemicals
Energy Production
Some decomposition reactions release energy we can use
Environmental Importance
Decomposition of organic waste in composting turns food scraps into nutrient-rich soil, reducing landfill waste!
Decomposition Reaction Quiz
Test your knowledge with this quick quiz! Choose the best answer for each question.
Frequently Asked Questions
Here are answers to common questions about decomposition reactions:
Decomposition Reaction Trivia
Discover some fascinating facts about decomposition reactions:
Rocket Science
Some rocket fuels use decomposition reactions! Ammonium perchlorate decomposes to produce gases that propel rockets into space.
Ancient Technology
Ancient Egyptians used thermal decomposition of limestone to make plaster for the pyramids over 4,500 years ago!
Nature's Cleanup Crew
In forests, decomposition reactions break down 90% of dead plant material, recycling nutrients back into the soil.
Flash Photography
Old flash photography used decomposition of magnesium powder to create bright flashes of light for photos.


