Ionization Energy - Definition, Examples, Quiz, FAQ, Trivia
Discover how atoms hold onto their electrons and what it takes to remove them!
What is Ionization Energy?
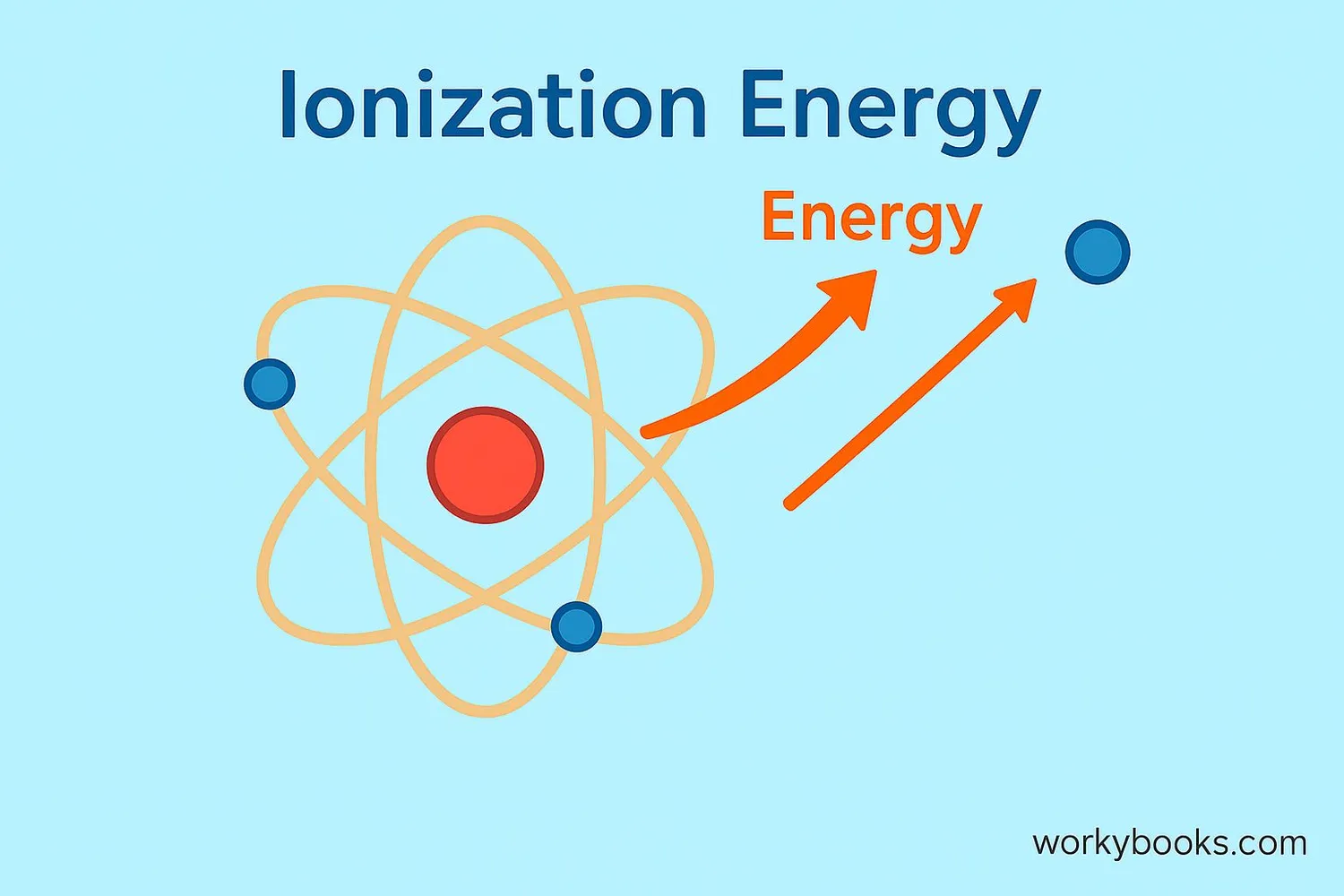
Ionization energy is the energy needed to remove an electron from an atom or molecule. Think of it like this:
Atoms are like tiny solar systems with electrons orbiting around a central nucleus. These electrons are held in place by electrical attraction. Ionization energy measures how tightly an atom holds onto its electrons - the higher the ionization energy, the harder it is to remove an electron!
Simple definition: Ionization energy is the "electron holding power" of an atom.
Science Fact!
The first ionization energy is the energy needed to remove the most loosely held electron from a neutral atom. After that, it takes more energy to remove additional electrons!
How Ionization Energy Works
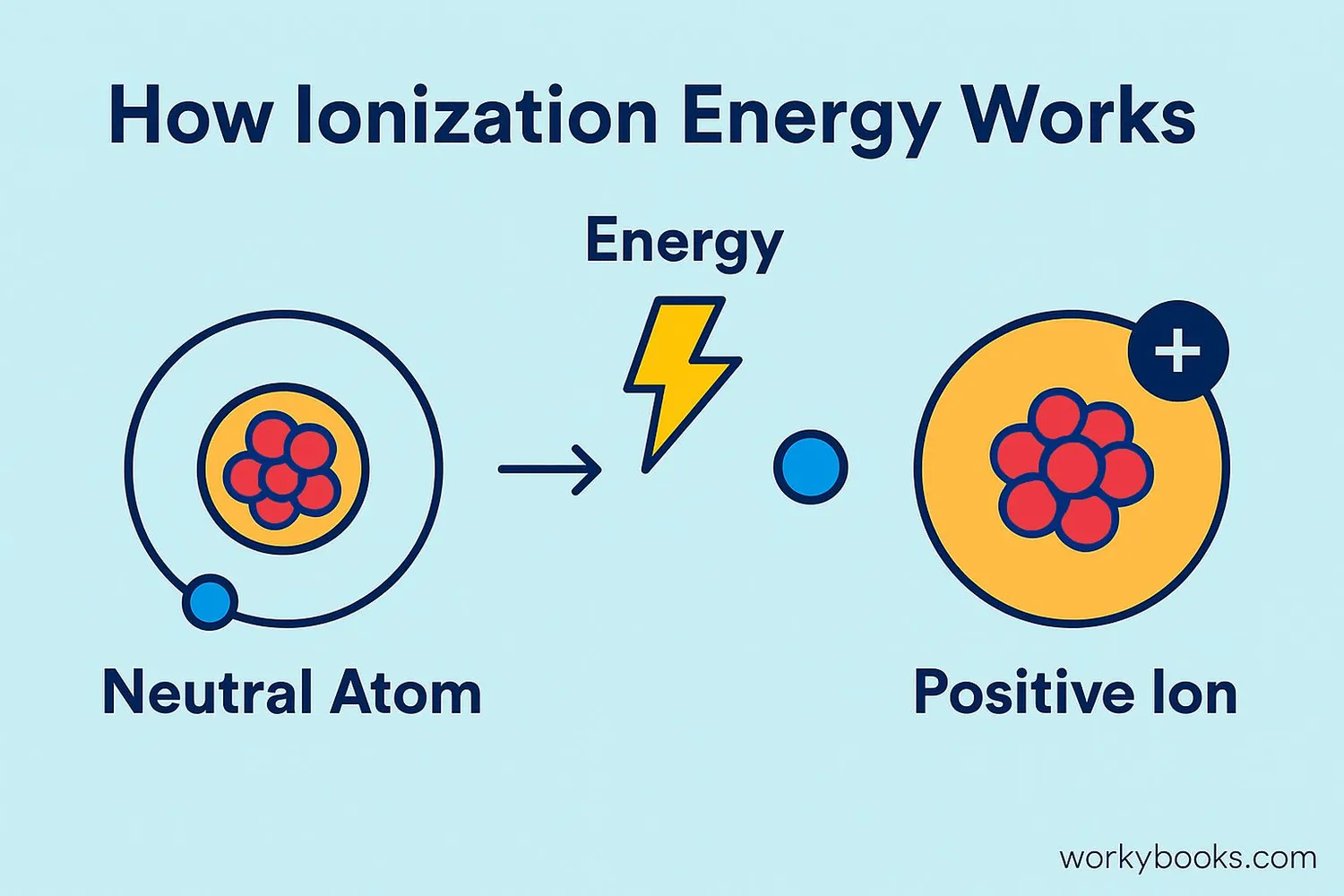
When we add energy to an atom (like heat or electricity), we can make an electron break free from its orbit. This process creates an ion - an atom with an electrical charge. Here's what happens:
Energy Added
Energy is supplied to the atom (heat, light, electricity)
Electron Removed
The outermost electron gains enough energy to escape
Ion Created
The atom becomes a positively charged ion (cation)
The ionization process can be written as:
Atom + Energy → Positive Ion + Electron
For example, when sodium loses an electron:
Na (sodium atom) + Energy → Na⁺ (sodium ion) + e⁻ (electron)
Atom Fact!
Noble gases like helium and neon have very high ionization energies because their electron configuration is very stable.
Factors Affecting Ionization Energy
Not all atoms hold their electrons equally tightly. Several factors affect ionization energy:
Atomic Size
Smaller atoms have higher ionization energy because electrons are closer to the nucleus
Nuclear Charge
Atoms with more protons have higher ionization energy (stronger pull on electrons)
Electron Shielding
Inner electrons shield outer electrons, reducing ionization energy
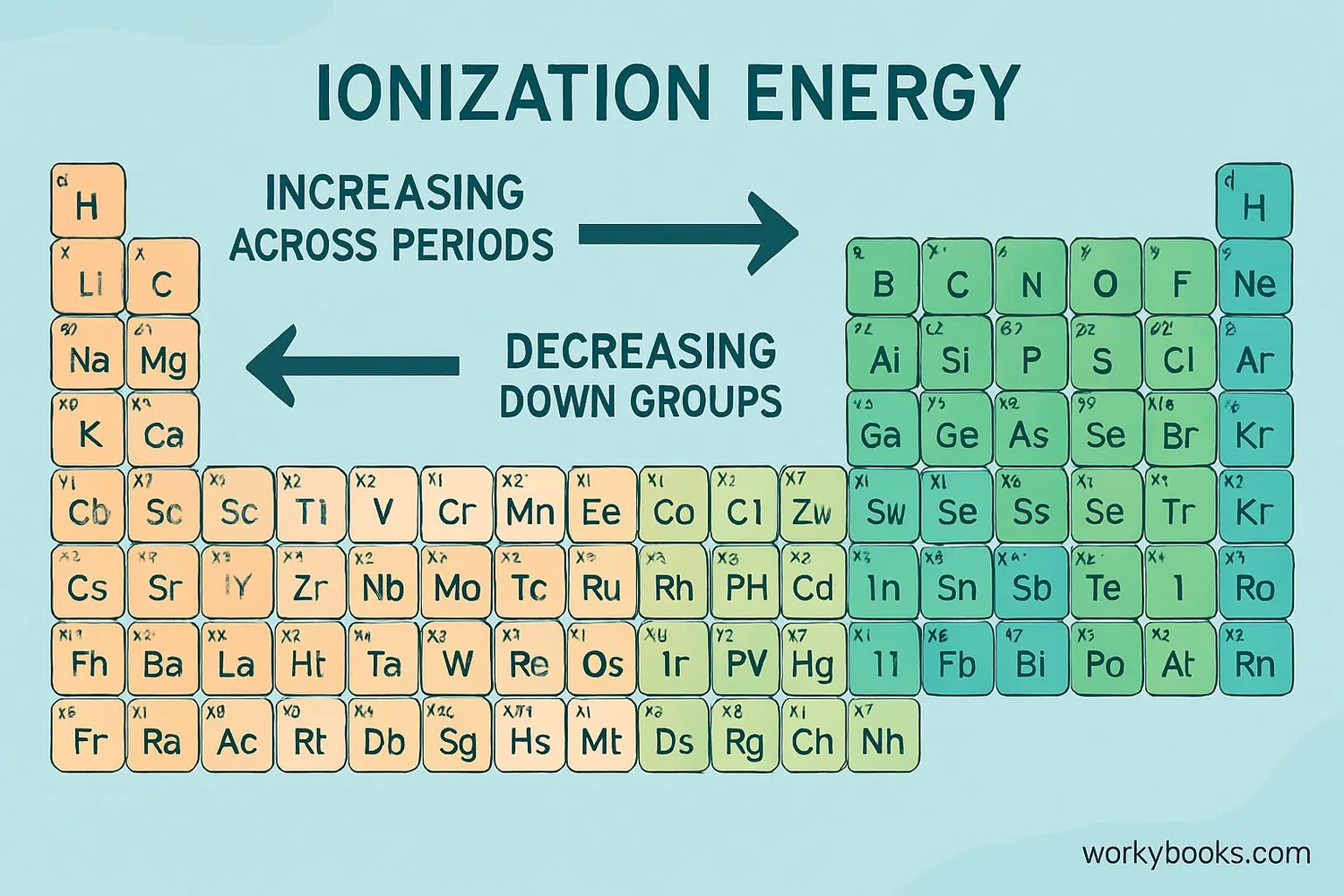
Periodic Table Trends:
• Across a period (left to right): Ionization energy increases
• Down a group (top to bottom): Ionization energy decreases
This is why alkali metals (Group 1) have the lowest ionization energies, while noble gases (Group 18) have the highest.
Why Ionization Energy is Important
Ionization energy helps us understand and predict how elements will behave:
Chemical Reactivity
Elements with low ionization energy are more reactive (like alkali metals)
Electrical Conductivity
Helps explain why some materials conduct electricity better than others
Technology Applications
Essential for understanding batteries, neon signs, and plasma screens
Scientists use ionization energy to:
• Predict how elements will form compounds
• Understand chemical bonding
• Develop new materials and technologies
• Explain why some elements are metals and others are non-metals
Ionization Energy Quiz
Test your knowledge with this ionization energy quiz! Answer all 5 questions to see how much you've learned.
Frequently Asked Questions
Here are answers to common questions about ionization energy:
Ionization Energy Trivia
Discover fascinating facts about ionization energy!
Energy Extreme
Helium has the highest first ionization energy of all elements at 2372 kJ/mol. That's equivalent to the energy of about 10,000 lightning bolts concentrated on a single atom!
The Gentle Giant
Francium has the lowest ionization energy of all naturally occurring elements. But it's so rare and radioactive that only about 20-30 grams exist on Earth at any time!
Technology Connection
Ionization energy principles are used in neon signs. Electricity provides energy to remove electrons from neon atoms, and when the electrons return, they emit colorful light!
Stellar Power
In stars, ionization happens constantly at extremely high temperatures. Our sun's surface is about 5,500°C - hot enough to ionize many elements and create plasma!


