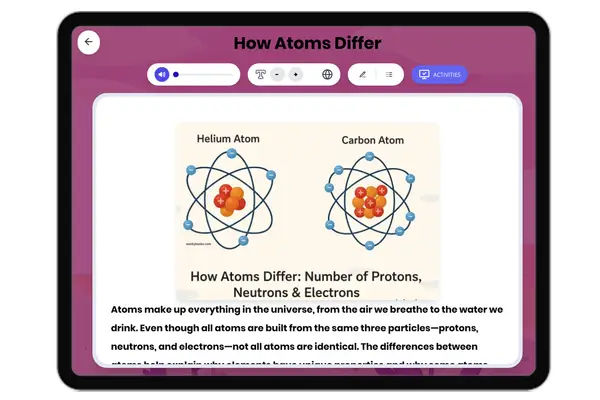
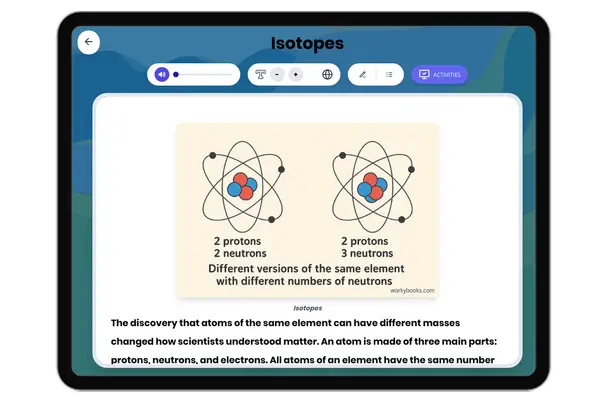
Nonmetals on Periodic Table
Interactive passage with audio narration, comprehension questions, and printable PDF.
Present
Open in classroom. No work savedAssign
Track progress per studentQuick play
Share with a link, no accountsDownload PDF
Print-ready, offline practice- Format
- Interactive (Online), Printable (PDF)
- Grades
- 5678
- Subjects
- sciencereadingela
- Standards
- MS-PS1-1
- Languages
- English, Spanish
What's included
Reading passage
Reading comprehension
Audio narration
With word word highlighting
Comprehension quiz
Auto-graded
Writing activity
Open-ended response
Glossary & flashcards
Vocabulary practice
Differentiated version
Adapted for varied levels
Spanish translation
Bilingual support
About this reader
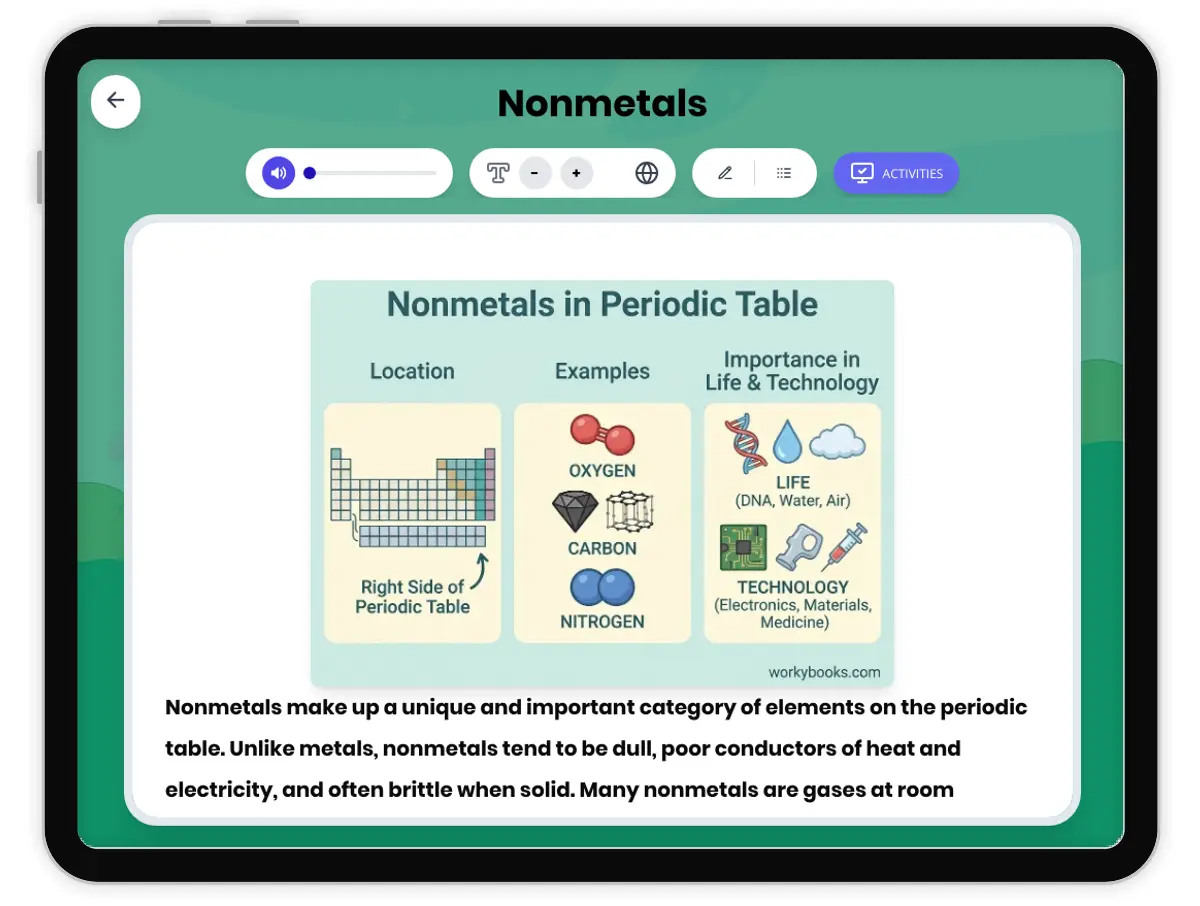
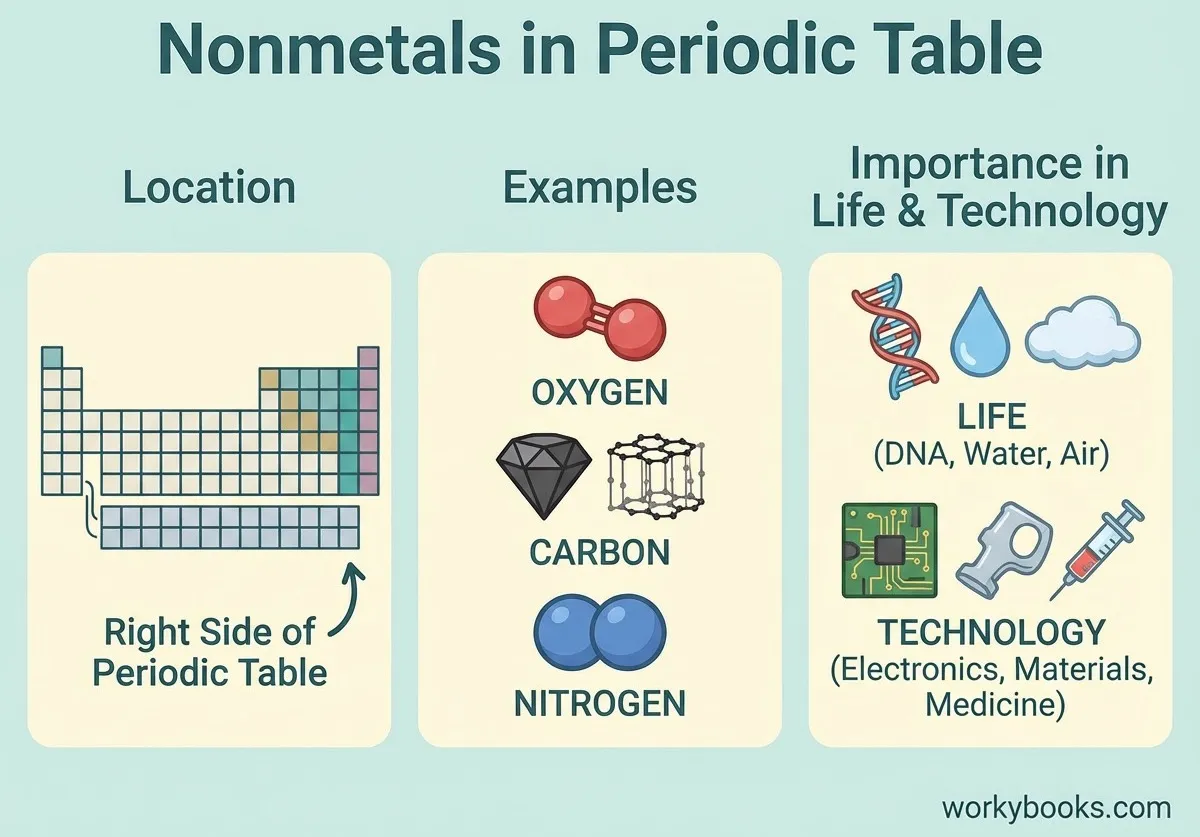
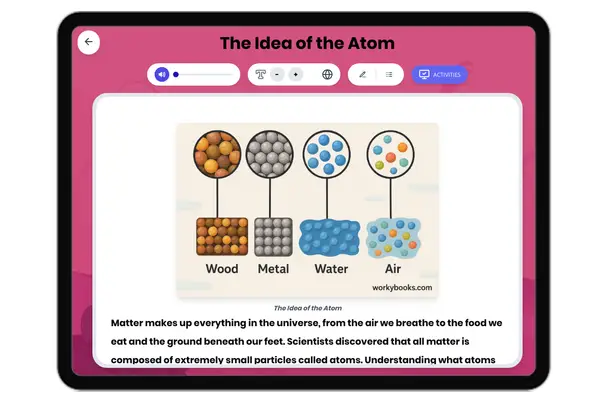
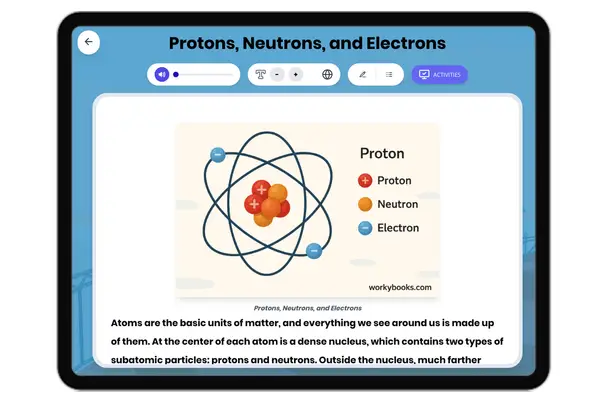
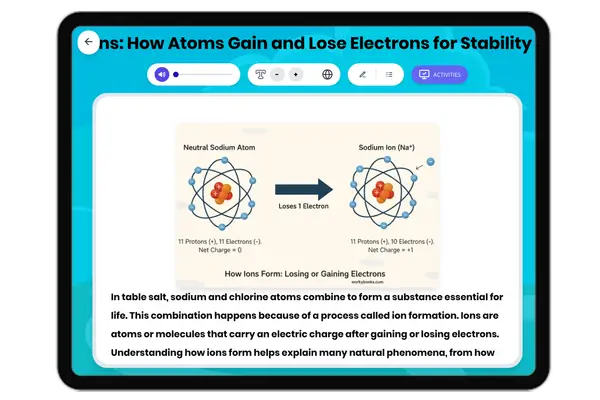
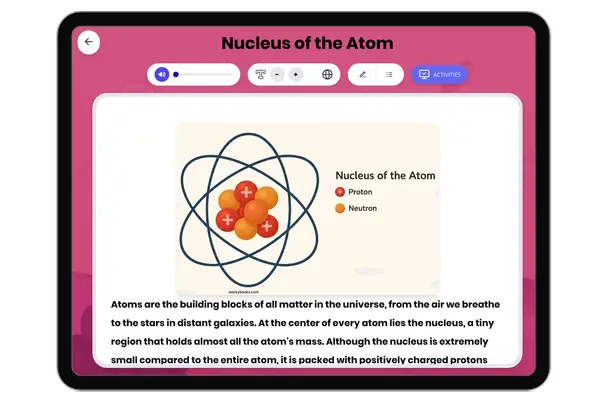
This NGSS-aligned passage for grades 6-8 introduces students to the fascinating world of nonmetals, a crucial category of elements found on the right side of the periodic table. The passage explores the unique properties of nonmetals, such as being dull, brittle, and poor conductors of electricity and heat. It uses real-world examples like oxygen, nitrogen, carbon, and sulfur to illustrate how nonmetals are essential for life, from the air we breathe to the structure of living things. Students will learn how nonmetals interact with other elements in systems and why their properties matter for biological, environmental, and technological processes. The activities include a glossary, differentiated readings, Spanish translations, quizzes, writing prompts, and graphic organizers, all designed for accessibility and deep understanding. The content is audio integrated, supporting diverse learning needs and making the passage ideal for classroom or independent study.
Written by Workybooks TeamPublished by Workybooks
Preview
Sample passage and quiz content
CONTENT PREVIEW
Who it's for
Perfect for the way you teach
Teachers
- Build comprehension skills
- Auto-graded quiz
- Differentiated reading
Parents
- Read together at home
- Improve fluency
- Quiet reading time
Homeschoolers
- Reading curriculum support
- Independent practice
- Track Lexile growth
Topics
nonmetalsperiodic tableoxygennitrogencarbonsulfursciencemiddle schoolNGSSpropertieselements